Textbook Question
Predict the product of the following Stille coupling reactions.
(b)
1193
views

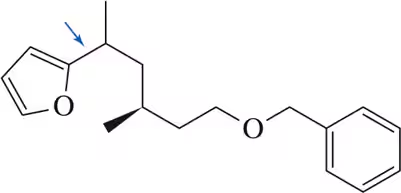
 Verified step by step guidance
Verified step by step guidance


Predict the product of the following Stille coupling reactions.
(b)
Predict the product of the following Negishi coupling reactions.
(b)
Predict the product of the following Stille coupling reactions.
(a)
Work backward to an appropriate organozinc halide and organohalide to make the bonds indicated by the blue arrows. There may be two possibilities for each.
(a)
Predict the product of the following Sonogashira coupling reactions.
(a)
Predict the product of the following Negishi coupling reactions.
(a)