Textbook Question
For the hydrogen(s) screened in blue, draw the signal you would expect to see in a ¹H NMR spectrum. At which chemical shift would the signal appear?
(c)
1046
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 15 - Structural Identification II: Nuclear Magnetic Resonance Spectroscopy
Ch. 15 - Structural Identification II: Nuclear Magnetic Resonance Spectroscopy Problem 61e
Problem 61e Verified step by step guidance
Verified step by step guidance


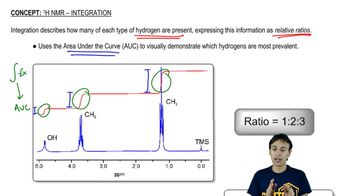
For the hydrogen(s) screened in blue, draw the signal you would expect to see in a ¹H NMR spectrum. At which chemical shift would the signal appear?
(c)
Draw the signal for the following multiplicities. What is the ratio of peaks within each signal?
(d) quintet
Draw the signal for the following multiplicities. What is the ratio of peaks within each signal?
(c) quartet
Draw the signal for the following multiplicities. What is the ratio of peaks within each signal?
(b) triplet
Draw the signal for the following multiplicities. What is the ratio of peaks within each signal?
(f) septet
For the hydrogen(s) screened in blue, draw the signal you would expect to see in a ¹H NMR spectrum. At which chemical shift would the signal appear?
(e)