Textbook Question
What product would be formed if the four-membered ring alcohol in Problem 70 were heated with an equivalent amount of HBr rather than with a catalytic amount of H2SO4?
907
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 10 - Reactions of Alcohols, Ethers, Epoxides, Amines, and Sulfur-Containing Compounds
Ch. 10 - Reactions of Alcohols, Ethers, Epoxides, Amines, and Sulfur-Containing Compounds Problem 70(8)
Problem 70(8) Verified step by step guidance
Verified step by step guidance


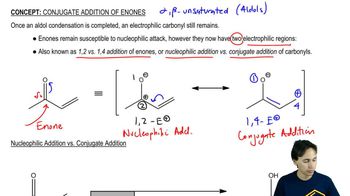
What product would be formed if the four-membered ring alcohol in Problem 70 were heated with an equivalent amount of HBr rather than with a catalytic amount of H2SO4?
Fill in each box with the appropriate reagent:
a.
Using the given starting material, any necessary inorganic reagents, and any carbon-containing compounds with no more than two carbons, indicate how the following syntheses could be carried out:
a.
Fill in each box with the appropriate reagent:
c.
Which of the following ethers would be obtained in greatest yield directly from alcohols?
Explain why (S)-2-butanol forms a racemic mixture when it is heated in sulfuric acid.