Textbook Question
Lawsone, found in the extract of henna, reacts with amino acid residues to give the characteristic color associated with henna tattoos. Suggest a mechanism for the reaction shown.
1024
views

 Mullins 1st Edition
Mullins 1st Edition Ch. 24 - Benzene II: Reactions Influenced by the Aromatic Ring
Ch. 24 - Benzene II: Reactions Influenced by the Aromatic Ring Problem 65
Problem 65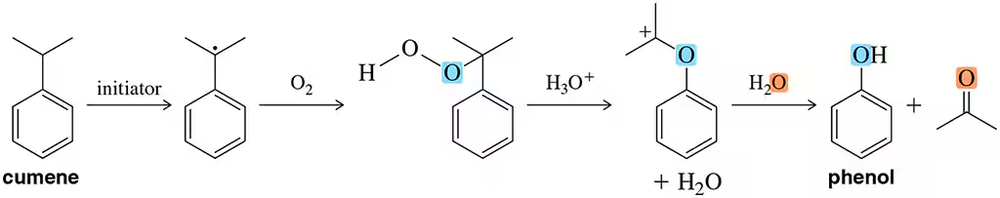
 Verified step by step guidance
Verified step by step guidance



Lawsone, found in the extract of henna, reacts with amino acid residues to give the characteristic color associated with henna tattoos. Suggest a mechanism for the reaction shown.
Synthesize the dye shown starting with aniline and naphthol using reactions from Chapters 23 and 24. [Hint. Start by identifying how each substituent might be installed and then plan the order of their introduction.]
Sulpiride is an antipsychotic. Provide the reagents (a) and (b) used to complete the synthesis.
Provide the reagents necessary to carry out the following synthesis. What is the purpose of steps (a) and (e)?