Crown ethers are able to solvate cations based on their size. Specifically, 15-crown-5 forms stable complexes with sodium. How would the addition of a crown ether change the rate of an SN2 reaction?
Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes

Mullins1st EditionOrganic Chemistry: A Learner Centered ApproachISBN: 9780137566471Not the one you use?Change textbook
All textbooks Mullins 1st Edition
Mullins 1st Edition Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes
Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes Problem 78
Problem 78
 Mullins 1st Edition
Mullins 1st Edition Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes
Ch. 12 - Substitution and Elimination: Reactions of Haloalkanes Problem 78
Problem 78Chapter 11, Problem 78
Of the three possible elimination mechanisms (Figure 12.50), this chapter focused on two of them (E1 and E2). The third possibility occurs in situations like the one below. What makes this mechanism favored under these conditions?
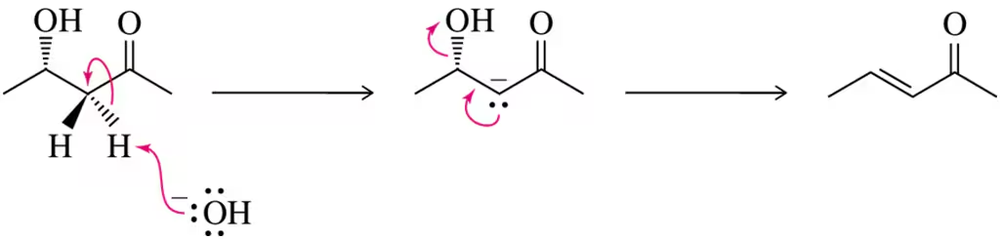
 Verified step by step guidance
Verified step by step guidance1
Understand the three elimination mechanisms: E1, E2, and E1cB. E1 involves a two-step process with carbocation formation, E2 is a concerted one-step process, and E1cB involves a carbanion intermediate. The problem hints at a third mechanism, likely E1cB, so we will focus on its conditions.
Analyze the given conditions. E1cB is favored when the leaving group is poor, the β-hydrogen is acidic (due to electron-withdrawing groups nearby), and the intermediate carbanion is stabilized by resonance or inductive effects.
Examine the structure of the substrate. Look for electron-withdrawing groups (e.g., -NO₂, -CN, -COOH) near the β-carbon, which can stabilize the carbanion intermediate through resonance or inductive effects.
Consider the reaction conditions. E1cB is typically favored in basic conditions, where a strong base can deprotonate the β-hydrogen to form the carbanion intermediate.
Conclude why this mechanism is favored. The combination of a poor leaving group, a stabilized carbanion intermediate, and basic conditions makes E1cB the preferred elimination pathway under these circumstances.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Video duration:
1mWas this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Elimination Mechanisms
Elimination mechanisms in organic chemistry refer to the processes by which atoms or groups are removed from a molecule, resulting in the formation of a double bond. The two primary types discussed are E1 (unimolecular elimination) and E2 (bimolecular elimination), each with distinct mechanisms and conditions. Understanding these mechanisms is crucial for predicting the products of reactions and the conditions under which they occur.
Recommended video:
Guided course

Intro to Substitution/Elimination Problems
E1 Mechanism
The E1 mechanism involves a two-step process where the leaving group departs first, forming a carbocation intermediate, followed by deprotonation to form a double bond. This mechanism is favored in polar protic solvents and with substrates that can stabilize the carbocation, such as tertiary alkyl halides. The rate of the reaction depends only on the concentration of the substrate, making it unimolecular.
Recommended video:
Guided course

Drawing the E1 Mechanism.
E2 Mechanism
The E2 mechanism is a concerted process where the leaving group and a hydrogen atom are removed simultaneously, resulting in the formation of a double bond. This mechanism requires a strong base and is favored in polar aprotic solvents. The reaction rate depends on both the substrate and the base, making it bimolecular, and it typically occurs with primary or secondary substrates where steric hindrance is less of a concern.
Recommended video:
Guided course

Drawing the E2 Mechanism.
Related Practice
Textbook Question
1263
views
Textbook Question
In this, and previous, chapters, we have seen 1,2-alkyl and 1,2-hydride shifts. If both are possible, as in the carbocation shown, which would you expect to occur? Explain your answer.
934
views
Textbook Question
The bromoalkanes shown below participate in SN1 reactions with the relative rates shown. Explain this trend. relative rate:
1137
views
Textbook Question
In Chapter 8, we learned about the chemistry of terpenes and the interesting reactions they can undergo. One such reaction is the acid-catalyzed conversion of nerol to terpineol. Suggest a mechanism for this transformation
.
1390
views
Textbook Question
Suggest a mechanism for the following reactions.
(c) Elimination:
1153
views