For each pair of compounds, predict the one with a higher boiling point. Which compounds have zero dipole moments?
c. cyclohexene or 1,2-dichlorocyclohexene

 Verified step by step guidance
Verified step by step guidance
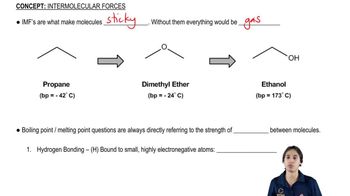

For each pair of compounds, predict the one with a higher boiling point. Which compounds have zero dipole moments?
c. cyclohexene or 1,2-dichlorocyclohexene
How many stereogenic double bonds are in octa-1,3,5-triene? How many stereocenters are there? Draw and name the four stereoisomers of octa-1,3,5-triene.
For each pair of compounds, predict the one with a higher boiling point. Which compounds have zero dipole moments?
b. cis- or trans-2,3-dichlorobut-2-ene
Draw the six stereoisomers of octa-2,4,6-triene. Explain why there are only six stereoisomers, rather than the eight we might expect for a compound with three stereogenic double bonds.
Teflon-coated frying pans routinely endure temperatures that would cause polyethylene or polypropylene to oxidize and decompose. Decomposition of polyethylene is initiated by free-radical abstraction of a hydrogen atom by O2. Bond-dissociation energies of C—H bonds are about 400 kJ/mol, and C—F bonds are about 460 kJ/mol. The BDE of the H—OO bond is about 192 kJ/mol, and the F—OO bond is about 63 kJ/mol. Show why Teflon (Figure 7-5) is much more resistant to oxidation than polyethylene is.
Use the data in Table 7-2 to predict the energy difference between 2,3-dimethylbut-1-ene and 2,3-dimethylbut-2-ene. Which of these double-bond isomers is more stable?