Show how you would accomplish the following syntheses. You may use whatever additional reagents you need.
(e)

 Verified step by step guidance
Verified step by step guidance


Show how you would accomplish the following syntheses. You may use whatever additional reagents you need.
(e)
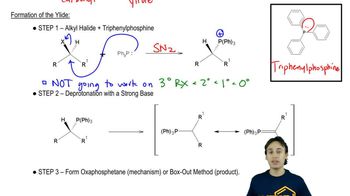
Like other strong nucleophiles, triphenylphosphine attacks and opens epoxides. The initial product (a betaine) quickly cyclizes to an oxaphosphetane that collapses to an alkene and triphenylphosphine oxide.
(b) Show how this sequence might be used to convert cis-cyclooctene to trans-cyclooctene.
Show how you would accomplish the following syntheses. You may use whatever additional reagents you need.
(d)
Show how you would accomplish the following syntheses. You may use whatever additional reagents you need.
(f)
Like other strong nucleophiles, triphenylphosphine attacks and opens epoxides. The initial product (a betaine) quickly cyclizes to an oxaphosphetane that collapses to an alkene and triphenylphosphine oxide.
(a) Show each step in the reaction of trans-2,3-epoxybutane with triphenylphosphine to give but-2-ene. What is the stereochemistry of the double bond in the product?
Write the sequence of steps required for the conversion of benzene into benzenediazonium chloride.