Textbook Question
(b) Do all of these ethers have common names?
1102
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 3 - An Introduction to Organic Compounds:Nomenclature, Physical Properties, and Structure
Ch. 3 - An Introduction to Organic Compounds:Nomenclature, Physical Properties, and Structure Problem 22a(1,2)
Problem 22a(1,2) Verified step by step guidance
Verified step by step guidance
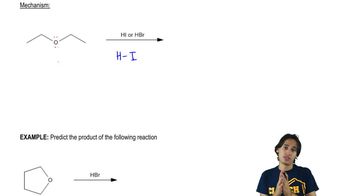


(b) Do all of these ethers have common names?
What is each ether’s systematic name?
3.
4.
Draw the structures and provide systematic names for parts a, b, and c by substituting a chlorine for a hydrogen of methylcyclohexane:
(c) three secondary alkyl halides
What is each ether's systematic name?
6.
What are their common names?
What is each ether's systematic name?
5.