The mechanism of glycoside formation is the same as the second part of the mechanism for acetal formation. Propose a mechanism for the formation of methyl β-D-glucopyranoside.

Predict the products obtained when d-galactose reacts with each reagent.(a) Br2 and H2O
 Verified step by step guidance
Verified step by step guidance
Verified video answer for a similar problem:
Key Concepts
D-Galactose Structure
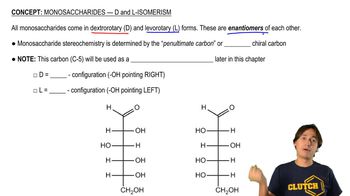
Oxidation Reactions

Halogenation of Sugars

Draw the structures of the compound ethyl β-D-ribofuranoside.
Allose is the C3 epimer of glucose, and ribose is the C2 epimer of arabinose.
Show the products that result from hydrolysis of amygdalin in dilute acid. Can you suggest why amygdalin might be toxic to tumor (and possibly other) cells?
Draw the structures of the compound α-D-allopyranose.
Allose is the C3 epimer of glucose, and ribose is the C2 epimer of arabinose.
Treatment of either anomer of fructose with excess ethanol in the presence of a trace of HCl gives a mixture of the α and β anomers of ethyl-D-fructofuranoside. Draw the starting materials, reagents, and products for this reaction. Circle the aglycone in each product.
Show the product that results when fructose is treated with an excess of methyl iodide and silver oxide.
