What product or products are obtained when D-galactose reacts with each of the following?
e. Br2 in water

 Verified step by step guidance
Verified step by step guidance
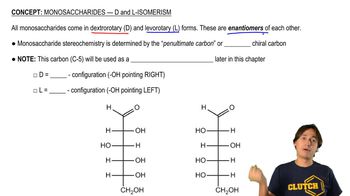


What product or products are obtained when D-galactose reacts with each of the following?
e. Br2 in water
Refer to Figure 20.5 to answer the following questions: b. People with type AB blood can receive blood from anyone, but they cannot give blood to everyone. To whom can they not give blood?
Refer to Figure 20.5 to answer the following questions: a. People with type O blood can donate blood to anyone, but they cannot receive blood from everyone. From whom can they not receive blood?
What product or products are obtained when d-galactose reacts with each of the following?
d. excess CH3I + Ag2O
What product or products are obtained when D-galactose reacts with each of the following?
c. NaBH4, followed by H3O+
Explain why the C-3 OH group of vitamin C is more acidic than the C-2 OH group.