Textbook Question
The first amino acid in the pentapeptide of Figure 26.50, Pyr, is not one of the 20 amino acids in Table 26.1. Based on the following structure of Pyr, from which natural amino acid is it likely derived?
778
views

 Verified step by step guidance
Verified step by step guidance
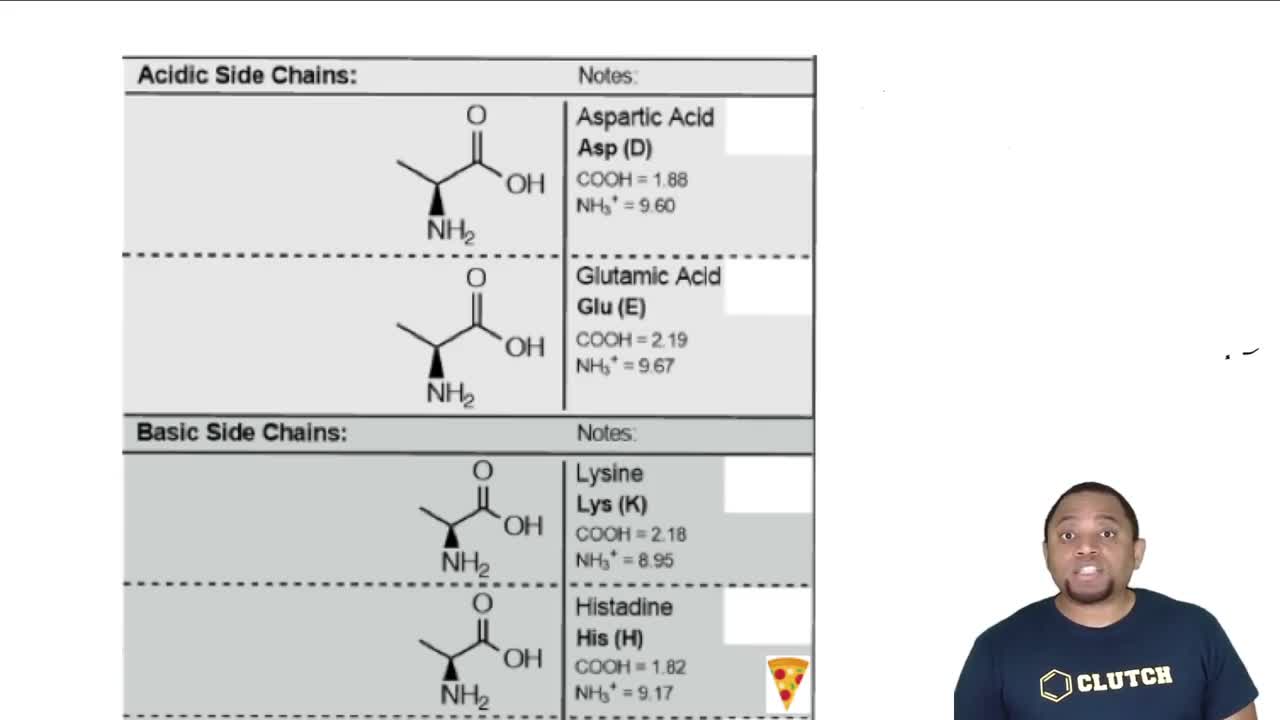

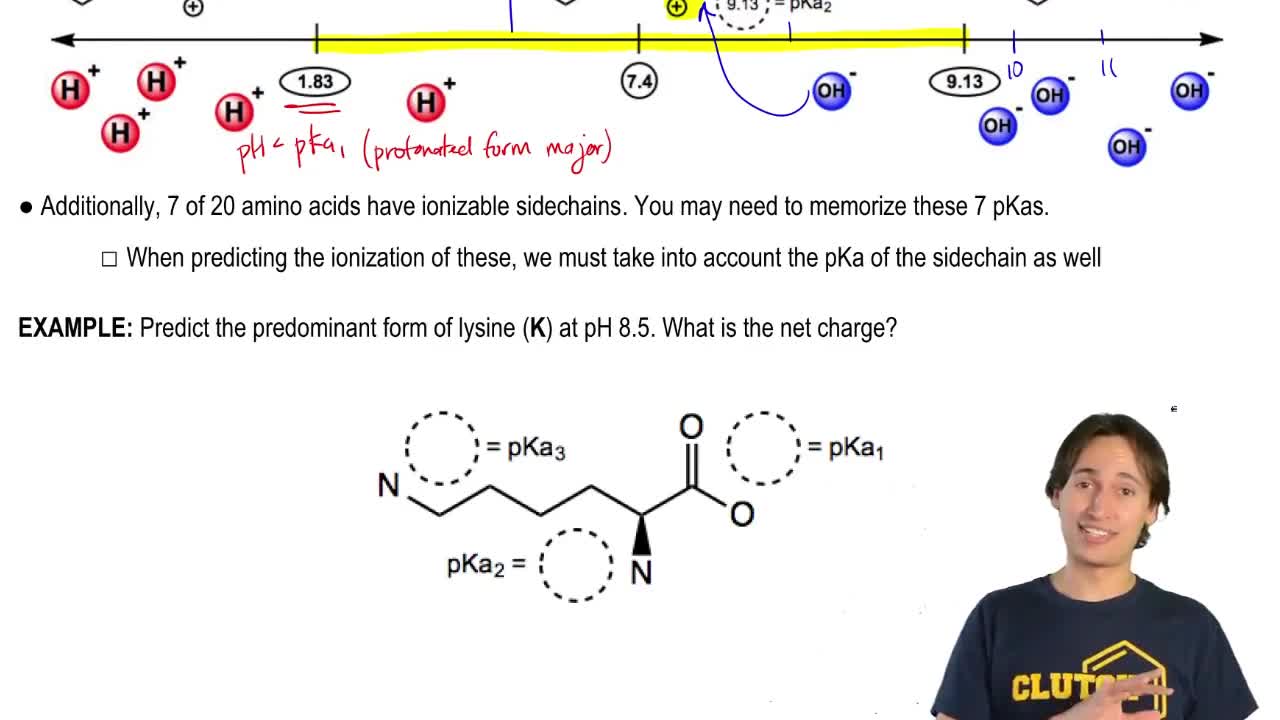
The first amino acid in the pentapeptide of Figure 26.50, Pyr, is not one of the 20 amino acids in Table 26.1. Based on the following structure of Pyr, from which natural amino acid is it likely derived?
Show how you would synthesize the following amino acids using the Strecker amino acid synthesis.
a. Ala
Why are conjugated molecules like the product of the ninhydrin reaction brightly colored?
Provide a mechanism for the protection of the amine as the benzylcarbamate shown in Figure 26.33(a). <IMAGE>