Textbook Question
Draw the structure of the predominant form of
(e) a mixture of alanine, lysine, and aspartic acid at (iii) pH 2.
800
views

 Verified step by step guidance
Verified step by step guidance


Draw the structure of the predominant form of
(e) a mixture of alanine, lysine, and aspartic acid at (iii) pH 2.
Draw the electrophoretic separation of Ala, Lys, and Asp at pH 9.7.
Draw the structure of the predominant form of
(d) glutamic acid at pH 7.
Draw the electrophoretic separation of Trp, Cys, and His at pH 6.0.
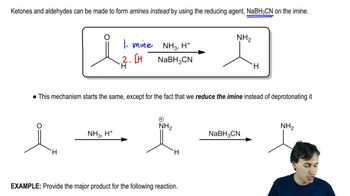
Show how the following amino acids might be formed in the laboratory by reductive amination of the appropriate α-ketoacid.
(a) alanine
Draw the structure of the predominant form of
(e) a mixture of alanine, lysine, and aspartic acid at (i) pH 6;