Textbook Question
Explain why the rate of bromination of methane decreases if HBr is added to the reaction mixture.
961
views


 Verified step by step guidance
Verified step by step guidance


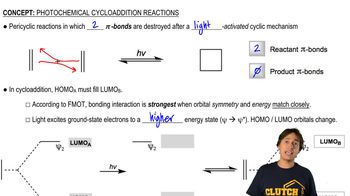
Explain why the rate of bromination of methane decreases if HBr is added to the reaction mixture.
Propose a mechanism for the following reaction:
A possible alternative mechanism to that shown in Problem 47 for the monochlorination of methane involves the following propagation steps:
How do you know that the reaction does not take place by this mechanism?
Draw the products of the following reactions, including all stereoisomers:
f.
Draw the products of the following reactions, including all stereoisomers:
e.
Draw the products of the following reactions, including all stereoisomers:
d.