Draw the two chair conformations of each compound, and label the substituents as axial and equatorial. In each case, determine which conformation is more stable.
c. cis-1-ethyl-3-methylcyclohexane
d. trans-1-ethyl-3-methylcyclohexane

 Verified step by step guidance
Verified step by step guidance
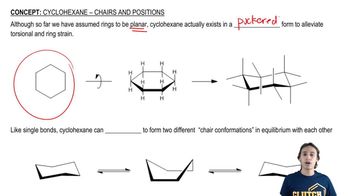


Draw the two chair conformations of each compound, and label the substituents as axial and equatorial. In each case, determine which conformation is more stable.
c. cis-1-ethyl-3-methylcyclohexane
d. trans-1-ethyl-3-methylcyclohexane
Draw the two chair conformations of each compound, and label the substituents as axial and equatorial. In each case, determine which conformation is more stable.
e. cis-1-ethyl-4-methylcyclohexane
f. trans-1-ethyl-4-methylcyclohexane
Use a Newman projection about the indicated bond to draw the most stable conformer for each compound.
a. 3-methylpentane about the C2―C3 bond
There are eight different five-carbon alkyl groups.
c. In each case, label the degree of substitution (primary, secondary, or tertiary) of the head carbon atom bonded to the main chain.
Use a Newman projection about the indicated bond to draw the most stable conformer for each compound.
b. 3,3-dimethylhexane about the C3―C4 bond
Draw the two chair conformations of each compound, and label the substituents as axial and equatorial. In each case, determine which conformation is more stable.
a. cis-1-ethyl-2-isopropylcyclohexane
b. trans-1-ethyl-2-isopropylcyclohexane