Textbook Question
For each of the following molecules, draw a 3-D representation.
(c) CHBr3
1211
views

 Verified step by step guidance
Verified step by step guidance



For each of the following molecules, draw a 3-D representation.
(c) CHBr3
For each of the following molecules, draw a 3-D representation.
(d) CHClBrI
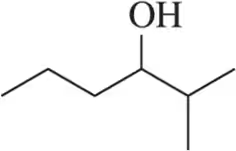
For each of the following molecules, identify all stereocenters and draw all possible stereoisomers.
(a)
For each of the following molecules, draw one 3-D representation at only the necessary atoms (that is, the ones with four different atoms/groups attached).
(c)
Identify all planes of symmetry in the following molecules/conformations, if any.
(f)
For each of the following molecules, draw one 3-D representation at only the necessary atoms (that is, the ones with four different atoms/groups attached).
(b)