Textbook Question
Explain why, when the guanidino group of arginine is protonated, the double-bonded nitrogen is the nitrogen that accepts the proton.
1153
views

 Verified step by step guidance
Verified step by step guidance


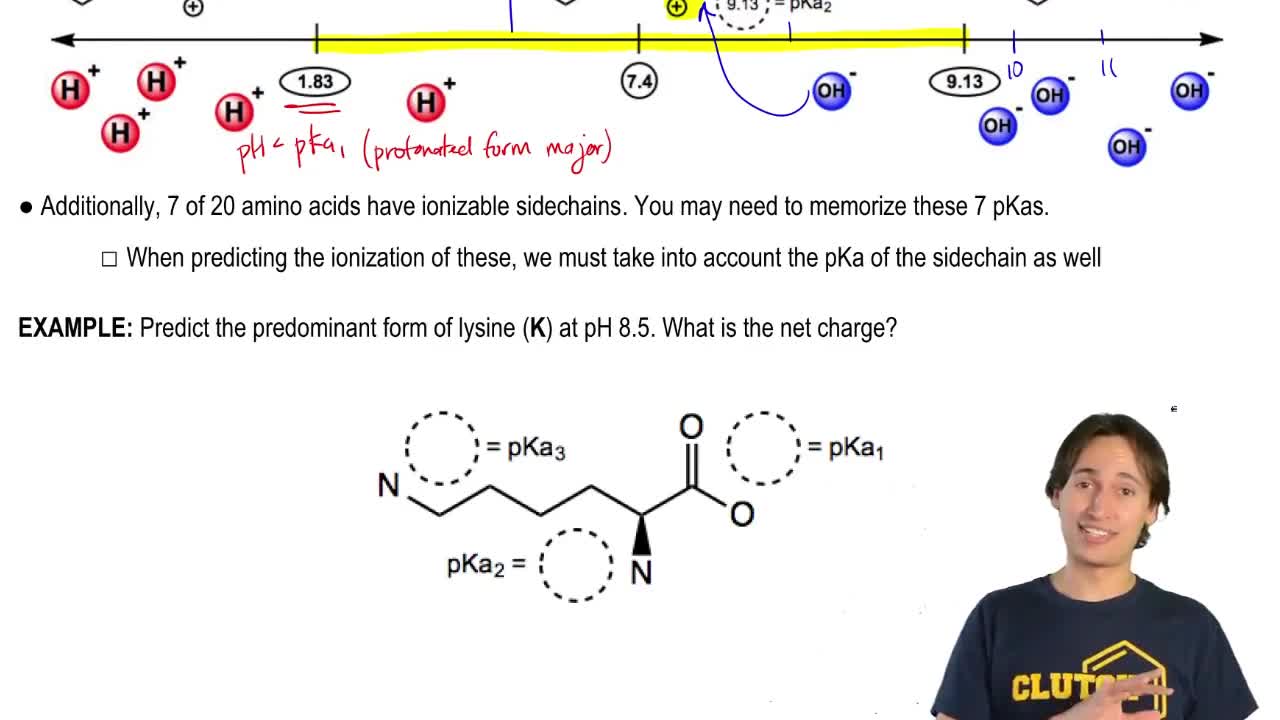
Explain why, when the guanidino group of arginine is protonated, the double-bonded nitrogen is the nitrogen that accepts the proton.
Explain why, when the imidazole ring of histidine is protonated, the double-bonded nitrogen is the nitrogen that accepts the proton.
Draw the predominant form for glutamate in a solution with the following
pH: b. 3
Draw the predominant form for glutamate in a solution with the following pH:
a. 0
Why is the pKa of the arginine side chain greater than the pKa of the lysine side chain?