Ch. 23 - The Organic Chemistry of the Coenzymes—Compounds Derived from Vitamins

All textbooks Bruice 8th Edition
Bruice 8th Edition Ch. 23 - The Organic Chemistry of the Coenzymes—Compounds Derived from Vitamins
Ch. 23 - The Organic Chemistry of the Coenzymes—Compounds Derived from Vitamins Problem 40c
Problem 40c
 Bruice 8th Edition
Bruice 8th Edition Ch. 23 - The Organic Chemistry of the Coenzymes—Compounds Derived from Vitamins
Ch. 23 - The Organic Chemistry of the Coenzymes—Compounds Derived from Vitamins Problem 40c
Problem 40cChapter 24, Problem 40c
Predict the major product of the following electrophilic aromatic substitution reactions.
(c) 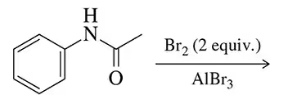
 Verified step by step guidance
Verified step by step guidance1
Identify the functional groups present in the molecule. The molecule contains an amide group attached to a benzene ring. The amide group is a moderate deactivating group and directs electrophilic substitution to the meta position.
Recognize the reagents used in the reaction. Bromine (Br₂) in the presence of aluminum bromide (AlBr₃) is a typical setup for bromination, an electrophilic aromatic substitution reaction.
Understand the role of AlBr₃. Aluminum bromide acts as a Lewis acid, facilitating the formation of the bromonium ion (Br⁺), which is the active electrophile in the reaction.
Determine the directing effects of the amide group. The amide group is a meta-directing group due to its electron-withdrawing nature, which means the bromine will likely add to the meta position relative to the amide group on the benzene ring.
Predict the major product. Given the conditions and the directing effects, the major product will be the bromination of the benzene ring at the meta position relative to the amide group. Since 2 equivalents of Br₂ are used, two bromine atoms will be added to the meta positions.

Verified video answer for a similar problem:
This video solution was recommended by our tutors as helpful for the problem above.
Was this helpful?
Key Concepts
Here are the essential concepts you must grasp in order to answer the question correctly.
Electrophilic Aromatic Substitution (EAS)
Electrophilic Aromatic Substitution is a reaction where an electrophile replaces a hydrogen atom on an aromatic ring. The aromatic ring acts as a nucleophile, and the reaction typically involves a catalyst to generate a more reactive electrophile. In this context, the benzene ring in the amide is the site of substitution, and the reaction conditions favor bromination.
Recommended video:
Guided course

EAS Review
Directing Effects of Substituents
Substituents on an aromatic ring influence the position where new groups are added during EAS. Electron-donating groups (EDGs) direct new substituents to ortho and para positions, while electron-withdrawing groups (EWGs) direct them to the meta position. The amide group in the given compound is an electron-withdrawing group, which typically directs electrophiles to the meta position.
Recommended video:

Directing Effects in Substituted Pyrroles, Furans, and Thiophenes Concept 1
Role of Lewis Acid Catalysts
Lewis acids, such as AlBr3, are used in EAS to enhance the electrophilicity of halogens like Br2. The Lewis acid forms a complex with the halogen, making it a more potent electrophile that can effectively attack the aromatic ring. This is crucial in facilitating the bromination of the benzene ring in the presence of the amide group.
Recommended video:

Acid-Base Catalysis Concept 3
0