The first step of a reaction called electrophilic aromatic substitution is as follows:
If this step is rate-determining for the overall reaction, which benzene derivative would you expect to react most quickly? Which would react most slowly?
(a)

 Mullins 1st Edition
Mullins 1st Edition Ch. 14 - Structural Identification I: Infrared Spectroscopy and Mass Spectrometry
Ch. 14 - Structural Identification I: Infrared Spectroscopy and Mass Spectrometry Problem 57
Problem 57
 Verified step by step guidance
Verified step by step guidance


The first step of a reaction called electrophilic aromatic substitution is as follows:
If this step is rate-determining for the overall reaction, which benzene derivative would you expect to react most quickly? Which would react most slowly?
(a)
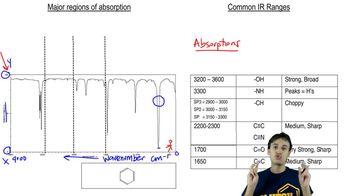
Would you expect the stretching band of the carbonyl to appear at a higher frequency for cyclohexanecarbaldehyde or benzaldehyde? Explain.
Justify the carbonyl stretching frequencies indicated for 3-chlorobenzaldehyde and 3-fluorobenzaldehyde.
Rank the following acetophenone derivatives on the basis of the carbonyl stretching frequency (1 = highest ; 4 = lowest ).
(a)
(b)
(c)
(d)
Under acidic conditions, alkene A can be isomerized to the more stable alkene B. How could IR spectroscopy be used to distinguish between A and B? [There are a few correct answers.]
Justify the carbonyl stretching frequencies indicated for benzaldehyde and 4-methoxybenzaldehyde.