Textbook Question
What reagents are needed to carry out the following syntheses?
1320
views

 Bruice 8th Edition
Bruice 8th Edition Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions
Ch. 6 - The Reactions of Alkenes • The Stereochemistry of Addition Reactions Problem 64c
Problem 64c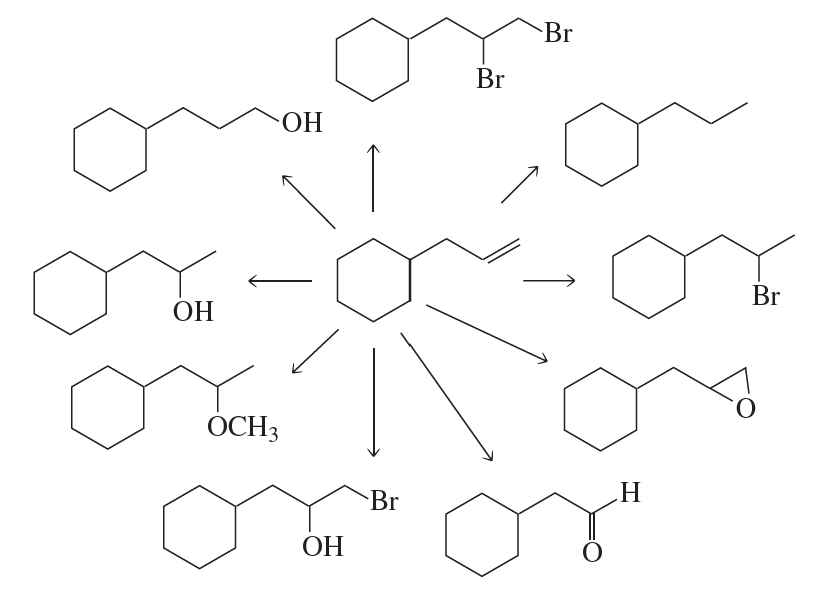
 Verified step by step guidance
Verified step by step guidance



What reagents are needed to carry out the following syntheses?
What is the major product of each of the following reactions?
a.
a. Identify two alkenes that react with HBr to form 1-bromo-1-methylcyclohexane without undergoing a carbocation rearrangement.
b. Would both alkenes form the same alkyl halide if DBr were used instead of HBr? (D is an isotope of H, so D+ reacts like H+.)
When 3-methyl-1-butene reacts with HBr, two alkyl halides are formed: 2-bromo-3-methylbutane and 2-bromo-2-methylbutane. Propose a mechanism that explains the formation of these two products.
What reagents are needed to carry out the following syntheses?
What reagents are needed to carry out the following syntheses?