Predict the products formed by sodium hydroxide-promoted dehydrohalogenation of the following compounds. In each case, predict which will be the major product.
d. cis-1-bromo-2-methylcyclohexane

 Verified step by step guidance
Verified step by step guidance

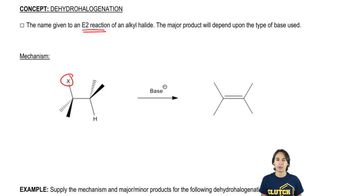

Predict the products formed by sodium hydroxide-promoted dehydrohalogenation of the following compounds. In each case, predict which will be the major product.
d. cis-1-bromo-2-methylcyclohexane
Predict the products formed by sodium hydroxide-promoted dehydrohalogenation of the following compounds. In each case, predict which will be the major product.
a. 1-bromobutane
b. 2-chlorobutane
c. 3-bromopentane
What halides would undergo E2 dehydrohalogenation to give the following pure alkenes?
d. methylenecyclohexane
Predict the major products of acid-catalyzed dehydration of the following alcohols.
(a)
(b)
Predict the products formed by sodium hydroxide-promoted dehydrohalogenation of the following compounds. In each case, predict which will be the major product.
e. trans-1-bromo-2-methylcyclohexane
What halides would undergo E2 dehydrohalogenation to give the following pure alkenes?
e. 4-methylcyclohexene