Predict the hybridization, geometry, and bond angles for the central atoms in
a. but-2-ene, CH3CH=CHCH3

 Verified step by step guidance
Verified step by step guidance
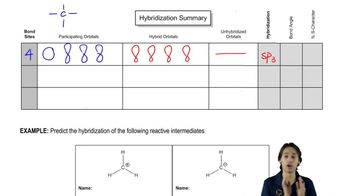
Predict the hybridization, geometry, and bond angles for the central atoms in
a. but-2-ene, CH3CH=CHCH3
Compute the empirical and molecular formulas for each of the following elemental analyses. In each case, propose at least one structure that fits the molecular formula.
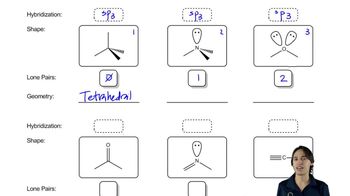
Predict the hybridization, geometry, and bond angles for the central atoms in
b. CH3CH=NH
The electrostatic potential maps for ammonia and water are shown here. The structure of ammonia is shown within its EPM. Note how the lone pair creates a region of high electron potential (red), and the hydrogens are in regions of low electron potential (blue). Show how your three-dimensional structure of water corresponds with its EPM.
Compute the empirical and molecular formulas for each of the following elemental analyses. In each case, propose at least one structure that fits the molecular formula.
Make a model of propane (C3H8), and draw this model using dashed lines and wedges to represent bonds going back and coming forward.