Some of the following compounds show aromatic properties, and others do not.
1. Predict which ones are likely to be aromatic, and explain why they are aromatic.
(n)
(o)

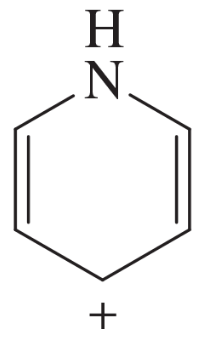
 Verified step by step guidance
Verified step by step guidance



Some of the following compounds show aromatic properties, and others do not.
1. Predict which ones are likely to be aromatic, and explain why they are aromatic.
(n)
(o)
Some of the following compounds show aromatic properties, and others do not.
1. Predict which ones are likely to be aromatic, and explain why they are aromatic.
2. Predict which nitrogen atoms are more basic than water and which are less basic.
(m)
Some of the following compounds show aromatic properties, and others do not.
1. Predict which ones are likely to be aromatic, and explain why they are aromatic.
2. Predict which nitrogen atoms are more basic than water and which are less basic.
(d)
Some of the following compounds show aromatic properties, and others do not.
1. Predict which ones are likely to be aromatic, and explain why they are aromatic.
2. Predict which nitrogen atoms are more basic than water and which are less basic.
(e)
The benzene ring alters the reactivity of a neighboring group in the benzylic position much as a double bond alters the reactivity of groups in the allylic position.
Benzylic cations, anions, and radicals are all more stable than simple alkyl intermediates.
a. Use resonance forms to show the delocalization (over four carbon atoms) of the negative charge of the benzyl anion.
Some of the following compounds show aromatic properties, and others do not.
1. Predict which ones are likely to be aromatic, and explain why they are aromatic.
(a)
(b)
(c)