 Back
BackAddition, Mechanisms, and Reactivity of Alkenes and Alkynes: A Comprehensive Study Guide
Study Guide - Smart Notes

Alkenes: Structure, Nomenclature, and Stereochemistry
Key Structural Vocabulary for Alkenes
Alkenes are hydrocarbons containing at least one carbon-carbon double bond. Their structure and nomenclature are foundational for understanding their reactivity and mechanisms.
Vinyl: A group directly attached to the double bond (C=C).
Allylic: A group adjacent to the double bond.
Vicinal: Substituents attached to adjacent carbons.
Geminal: Substituents attached to the same carbon.
Substitution Pattern: Classified as mono-, di-, tri-, or tetra-substituted based on the number of non-hydrogen groups attached to the double bond.
Isolated, Conjugated, Cumulated: Describes the relative positions of multiple double bonds.
E and Z Configurations of Alkenes
The stereochemistry of alkenes is described using the E/Z system, especially for tri- and tetra-substituted alkenes where cis/trans is insufficient. The Cahn-Ingold-Prelog (CIP) priority rules are used:
Z (zusammen): Higher priority groups on each carbon are on the same side.
E (entgegen): Higher priority groups are on opposite sides.
For cyclic alkenes, rings smaller than seven members cannot maintain E/Z geometry due to ring strain.
Reactivity and Mechanisms of Alkenes
Reactivity Trends
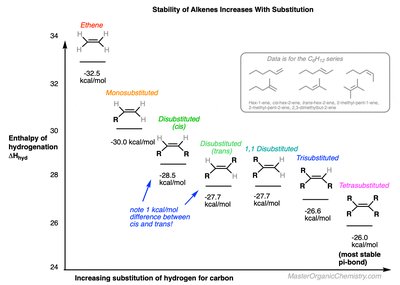
Alkenes undergo addition reactions, where the π bond is converted into two new σ bonds. Reactivity decreases with increased substitution and conjugation due to stabilization of the π bond. Enthalpy of hydrogenation is used to compare reactivity: more negative values indicate higher reactivity.
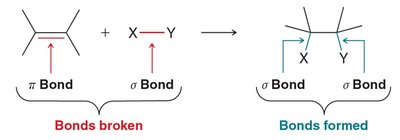
General Mechanism: Electrophilic Addition
Alkenes act as nucleophiles, attacking electrophilic species. The mechanism depends on the nature of the electrophile:
Hard Electrophiles: Small, highly charged, often produce carbocation intermediates (stepwise, random stereochemistry).
Soft Electrophiles: Larger, polarizable, often produce cyclic intermediates (concerted, syn addition).
The outcome is determined by the intermediate formed, which dictates both regiochemistry and stereochemistry.
Key Definitions of Reaction Outcomes
Markovnikov Product: The hydrogen adds to the carbon with more hydrogens ("the rich get richer").
Anti-Markovnikov Product: The hydrogen adds to the less substituted carbon, typically in the presence of peroxides or via radical mechanisms.
Syn Addition: Both groups add to the same face of the double bond.
Anti Addition: Groups add to opposite faces of the double bond.
Random Addition: Both syn and anti products are formed, usually via carbocation intermediates.
Summary Table: Alkene Addition Reactions
# | Reaction | Electrophile | Intermediate | Product | Mode of Addition |
|---|---|---|---|---|---|
1 | Hydrohalogenation | HX | Carbocation | Alkyl halide | Random, Markovnikov |
2 | Acid-catalyzed Hydration | H3O+ | Carbocation | Alcohol | Random, Markovnikov |
3 | Hydroboration-Oxidation | BH3 | Trialkylborane | Alcohol | Syn, Anti-Markovnikov |
4 | Oxymercuration-Demercuration | Hg(OAc)2 | Mercurinium ion | Alcohol | Anti, Markovnikov |
5 | Halogenation | X2 | Halonium ion | Dihalide | Anti |
6 | Hydrohalogenation (Radical) | HX + ROOR | Radical | Alkyl halide | Random, Anti-Markovnikov |
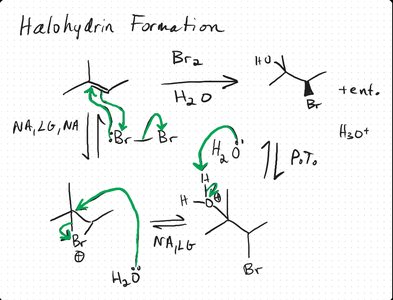
7 | Halohydrin Formation | X2 + H2O | Halonium ion | Halohydrin | Anti |
8 | Hydrogenation | H2 + metal | Metal complex | Alkane | Syn |
9 | Dihydroxylation | OsO4 | Cyclic osmate | Diol | Syn |
10 | Epoxidation | RCO3H | Epoxide | Epoxide | Syn |
11 | Ozonolysis | O3 | Ozonide | Aldehyde/Ketone | N/A |
Mechanisms of Key Alkene Reactions
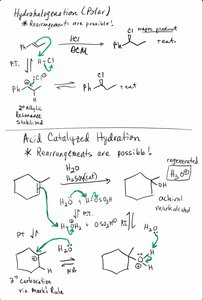
Hydrohalogenation and Acid-Catalyzed Hydration
Both reactions proceed via carbocation intermediates, allowing for rearrangements and leading to Markovnikov products. Acid-catalyzed hydration uses water and an acid catalyst to add H and OH across the double bond.
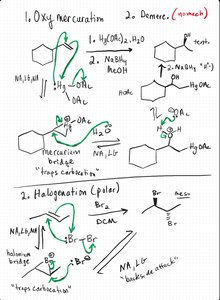
Oxymercuration-Demercuration and Halogenation
Oxymercuration avoids carbocation rearrangements by forming a mercurinium ion intermediate, leading to Markovnikov alcohols. Halogenation forms a halonium ion, resulting in anti addition of halogens across the double bond.
Halohydrin Formation
When alkenes react with halogens in water, a halohydrin is formed via anti addition. The mechanism involves a halonium ion intermediate, followed by nucleophilic attack by water.
Alkynes: Structure, Reactivity, and Mechanisms
Internal vs. Terminal Alkynes
Alkynes are classified as internal (triple bond between two carbons) or terminal (triple bond at the end of the chain). Terminal alkynes have an acidic proton and can be deprotonated to form alkynide ions.
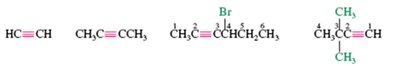
Alkyne Addition Mechanisms
Alkynes undergo similar addition reactions as alkenes, but with some differences:
Hydroboration and oxymercuration conditions differ due to the reactivity of alkynes.
Alkynes can react via bimolecular (AdE2) or termolecular (AdE3) mechanisms.
Regiochemistry generally follows Markovnikov's rule.
Alkyne Formation
Alkynes are synthesized from dihalides via double elimination, typically using strong bases such as NaNH2 or LDA. Terminal alkynes require excess base and a water work-up.
Redox in Organic Chemistry
Definitions
Reduction: Gain of electrons, gain of hydrogen, or loss of oxygen.
Oxidation: Loss of electrons, loss of hydrogen, or gain of oxygen.
Oxidation State: Reflects electron ownership in bonds; carbon can range from -4 to +4.
Radical Reactions
Properties and Mechanisms
Radicals are species with an unpaired electron. They are less reactive than carbocations and can undergo rearrangements and resonance stabilization. Radical mechanisms proceed in three phases:
Initiation: Homolytic cleavage generates radicals.
Propagation: Radicals react to form new radicals, sustaining the chain reaction.
Termination: Two radicals combine, ending the chain.

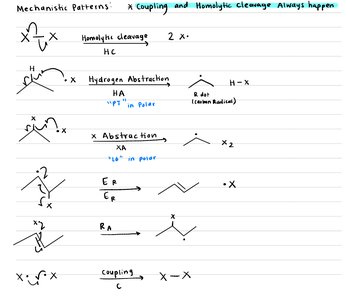
Radical Halogenation
Alkanes can be functionalized via radical halogenation. Selectivity depends on the type of C-H bond and the halogen used (bromination is more selective than chlorination).
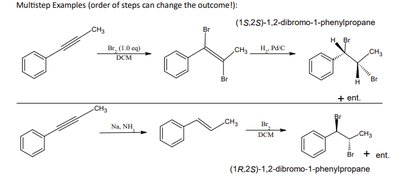
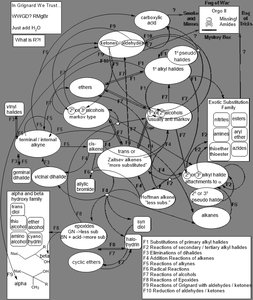
Synthetic Roadmap and Multistep Synthesis
Roadmap Overview
The synthetic roadmap helps visualize possible transformations between functional groups. One-step and two-step transformations are common, and the order of steps can affect the outcome, especially in stereochemistry.
Summary Table: Geometry and Intermediates
Intermediate | Geometry | Orbital | Electron Density |
|---|---|---|---|
Carbocation | Trigonal planar | Empty p | Deficient |
Radical | Approx. trigonal planar | Singly occupied p | Deficient (varies) |
Carbanion | Trigonal pyramidal | sp3 | Rich |
Additional info: This guide integrates key mechanistic details, stereochemical outcomes, and synthetic strategies for alkenes and alkynes, providing a comprehensive resource for exam preparation and conceptual understanding.
