 Back
BackAlcohols and Phenols: Structure, Properties, Synthesis, and Reactions
Study Guide - Smart Notes

Alcohols and Phenols
Introduction to Alcohols and Phenols
Alcohols and phenols are important classes of organic compounds characterized by the presence of a hydroxyl group (–OH). Their structural features, physical and chemical properties, and methods of synthesis are fundamental to organic chemistry and pharmaceutical sciences.
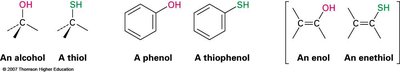
Alcohols: Compounds where the –OH group is bonded to a saturated, sp3-hybridized carbon atom.
Phenols: Compounds where the –OH group is bonded directly to an aromatic ring.
Enols: Compounds with the –OH group bonded to a vinylic, sp2-hybridized carbon.
Occurrence and Applications
Alcohols and phenols are widely distributed in nature and have significant industrial and pharmaceutical applications.
Methanol (wood alcohol): Used industrially but toxic to humans.
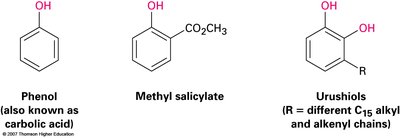
Phenols: Found in disinfectants (e.g., Dettol, TCP), flavoring agents (e.g., methyl salicylate), and as intermediates in synthesis.

Structural Features and Classification
Classification of Alcohols
Alcohols are classified based on the number of organic groups attached to the carbon bearing the hydroxyl group:
Primary (1°) alcohol: One organic group attached.
Secondary (2°) alcohol: Two organic groups attached.
Tertiary (3°) alcohol: Three organic groups attached.
Example structures:
Ethanol (primary), 2-propanol (secondary), 2-methyl-2-propanol (tertiary).
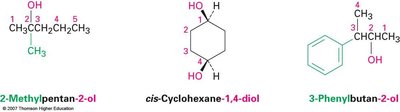
Naming Alcohols and Phenols (IUPAC)
The IUPAC system names alcohols as derivatives of the parent alkane, replacing the –e ending with –ol. The chain is numbered to give the –OH group the lowest possible number.
Select the longest carbon chain containing the –OH group.
Number the chain from the end nearest the –OH group.
Name and number substituents, listing them alphabetically.
Physical Properties of Alcohols and Phenols
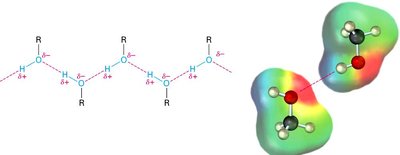
Hydrogen Bonding and Solubility
The O—H bond in alcohols and phenols is polar, allowing for hydrogen bonding with water and other alcohols. This confers hydrophilicity and affects solubility and boiling points.
Water solubility decreases as the non-polar (hydrocarbon) portion increases.
Salts of alcohols and phenols are more water-soluble due to ion-dipole interactions.
Alcohols and phenols have higher boiling points than similar-sized hydrocarbons due to hydrogen bonding.
Chemical Properties: Acidity and Basicity
Acid-Base Behavior
Alcohols and phenols are both weak acids and weak bases:
As weak acids, they do not react with weak bases (e.g., amines, bicarbonate).
As weak bases, they can be protonated by strong acids to yield oxonium ions (ROH2+).
They react with alkali metals and strong bases (e.g., NaH, NaNH2).
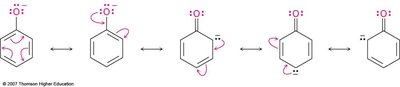
Acidity of Alcohols and Phenols
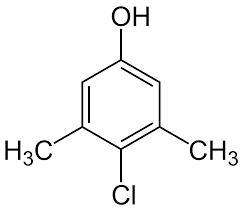
The acidity depends on the O—H bond strength and the stability of the conjugate base. Phenols are more acidic than alcohols due to resonance stabilization of the phenoxide ion.
Electron-withdrawing substituents increase acidity by stabilizing the negative charge.
Electron-donating substituents decrease acidity by concentrating the negative charge.
Synthesis of Alcohols
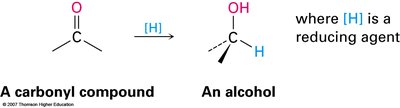
Reduction of Carbonyl Compounds
The most common method for synthesizing alcohols is the reduction of carbonyl compounds (aldehydes, ketones, carboxylic acids, esters).
Aldehydes are reduced to primary alcohols.
Ketones are reduced to secondary alcohols.
Carboxylic acids and esters are reduced to primary alcohols (usually with LiAlH4).

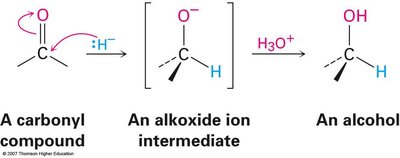
Mechanism of Carbonyl Reduction
The reduction involves nucleophilic addition of a hydride ion (H–) to the electrophilic carbonyl carbon, forming an alkoxide intermediate, which is then protonated to yield the alcohol.
Common reducing agents: NaBH4 (for aldehydes/ketones), LiAlH4 (for acids/esters).
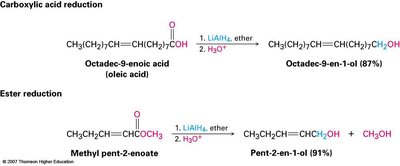
Reduction of Carboxylic Acids and Esters
Carboxylic acids and esters are reduced to primary alcohols, typically using lithium aluminium hydride (LiAlH4).
Two hydrogens are added to the carbonyl carbon during reduction.
Reactions of Alcohols
Dehydration, Substitution, and Oxidation
Alcohols can undergo several important reactions:
Dehydration: Forms alkenes.
Conversion to Alkyl Halides: Using SOCl2, PCl5, or PBr3.
Oxidation: Primary alcohols yield aldehydes or carboxylic acids; secondary alcohols yield ketones; tertiary alcohols are resistant to oxidation.
Esterification: Reaction with carboxylic acids to form esters.
Ether Formation: Reaction with alkyl halides to form ethers.
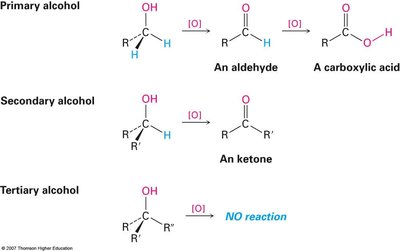
Summary Table: Oxidation of Alcohols
Alcohol Type | Oxidation Product |
|---|---|
Primary (1°) | Aldehyde (mild), Carboxylic acid (strong) |
Secondary (2°) | Ketone |
Tertiary (3°) | No reaction under normal conditions |
Conclusion
Understanding the structure, properties, synthesis, and reactions of alcohols and phenols is essential for mastering organic chemistry. Their behavior as weak acids and bases, their ability to form hydrogen bonds, and their versatile reactivity make them central to both biological and industrial chemistry.
