 Back
BackAldehydes and Ketones: Structure, Properties, and Reactions
Study Guide - Smart Notes

Chapter 18: Aldehydes and Ketones
18.1 Carbonyl Compounds
The carbonyl group (C=O) is a defining feature of several important classes of organic compounds, including aldehydes and ketones. The carbonyl carbon is sp2 hybridized, resulting in a planar structure and significant reactivity due to the polarization of the C=O bond.
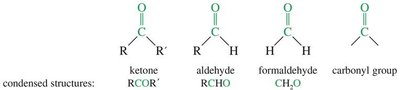
Aldehydes: The carbonyl carbon is bonded to at least one hydrogen atom.
Ketones: The carbonyl carbon is bonded to two carbon atoms.

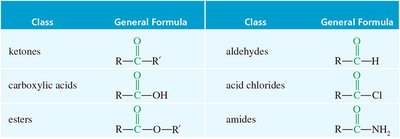
Other classes of carbonyl compounds include carboxylic acids, esters, acid chlorides, and amides, all of which contain the C=O group but differ in their substituents.
Condensed structures are often used to represent these compounds for simplicity.
18.2 Structure of the Carbonyl Group
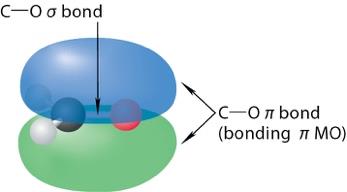
The carbonyl group consists of a sigma (σ) bond and a pi (π) bond between carbon and oxygen. The π bond arises from the sideways overlap of p orbitals, while the σ bond is formed by head-on overlap.
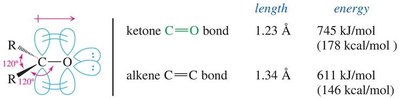
The C=O bond is shorter (1.23 Å) and stronger (745 kJ/mol) than the C=C bond in alkenes (1.34 Å, 611 kJ/mol).
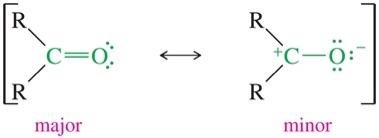
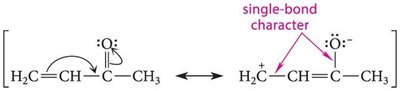
The bond is highly polar due to the electronegativity difference between carbon and oxygen, resulting in a partial positive charge on carbon and a partial negative charge on oxygen.
The polarity of the carbonyl group is reflected in the dipole moments of aldehydes and ketones, which are higher than those of similar molecules without the C=O group.

Resonance structures can be drawn for the carbonyl group, but the neutral form is the major contributor due to complete octets and lack of formal charges.
18.3 Nomenclature of Ketones and Aldehydes
Naming of aldehydes and ketones follows IUPAC rules, with some common names still in use.
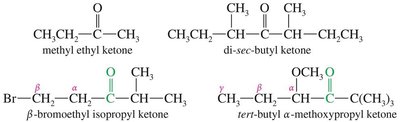
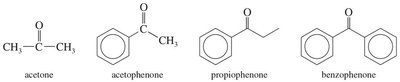
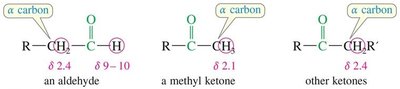
Ketones (Common Names): Named as alkyl attachments to the C=O, using Greek letters (α, β, γ) to indicate positions.
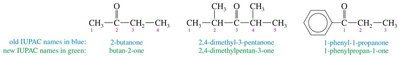
Ketones (IUPAC Names): The parent chain includes the carbonyl carbon; the suffix is changed from "-e" to "-one". For cyclic ketones, the carbonyl is always position 1.
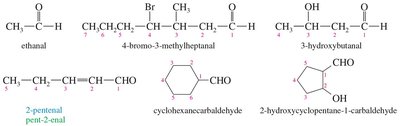
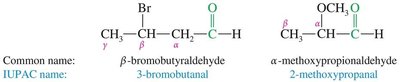
Aldehydes (IUPAC Names): The parent chain includes the carbonyl carbon, which is always position 1; the suffix is changed from "-e" to "-al". If attached to a ring, use the suffix "-carbaldehyde".
When other functional groups are present, the priority order is: acids > esters > aldehydes > ketones > alcohols > amines > alkenes/alkynes > ethers > halides.
Common names for acids and aldehydes are often derived from Latin or Greek roots, reflecting their natural sources or properties.
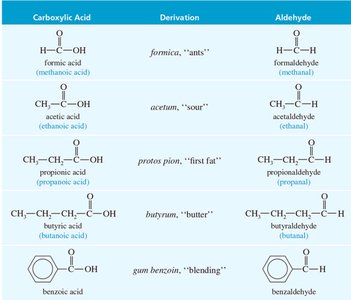
Some aromatic ketones have historical common names.
Common and IUPAC names for substituted aldehydes are also used.
18.4 Physical Properties of Ketones and Aldehydes
Aldehydes and ketones are generally liquids at room temperature. Their boiling points are lower than those of alcohols due to the absence of hydrogen-bond donors, but higher than those of alkanes and ethers of similar molecular weight.
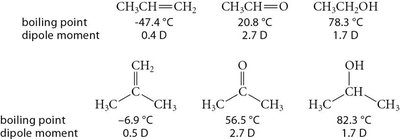
They are good solvents for alcohols and can accept hydrogen bonds from O–H or N–H groups.
Acetone and acetaldehyde are miscible with water.
Formaldehyde is a gas at room temperature; its aqueous solution is called formalin.
18.5 Spectroscopy of Ketones and Aldehydes
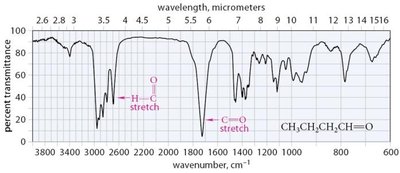
IR Spectroscopy
Strong C=O stretch at 1710 cm-1 for ketones and 1725 cm-1 for simple aldehydes.
Aldehydes show characteristic C–H stretching near 2710 cm-1 and 2810 cm-1.
Conjugation lowers the absorption frequency; ring strain increases it.
1H NMR Spectroscopy
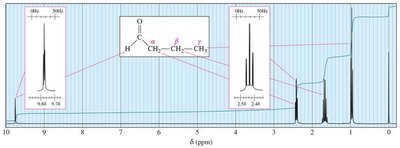
Aldehyde protons appear at δ 9–10 ppm.
Alpha protons (adjacent to the carbonyl) appear at δ 2.1–2.4 ppm.
Protons further from the carbonyl appear at lower δ values.
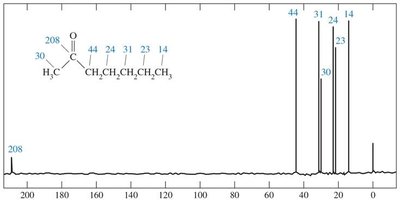
13C NMR Spectroscopy
Carbonyl carbons appear at δ 180–220 ppm.
Alpha carbons appear at δ 30–50 ppm.
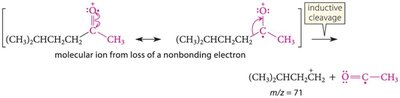
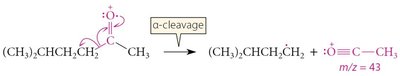
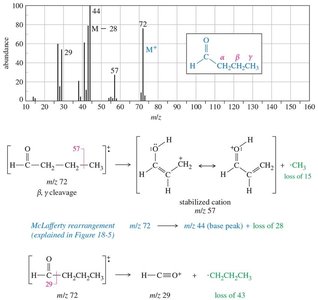
Mass Spectrometry
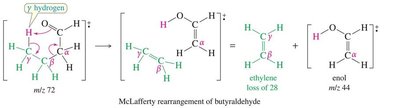
Characteristic peaks arise from inductive and alpha cleavage.
McLafferty rearrangement involves a γ-hydrogen transfer, forming an alkene and an enol.
UV/VIS Spectroscopy
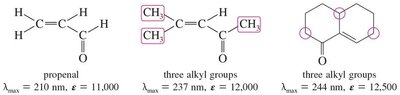
Conjugated carbonyl systems show π → π* absorption in the UV region.
Additional conjugation increases λmax by about 30 nm; additional alkyl groups increase it by about 10 nm.
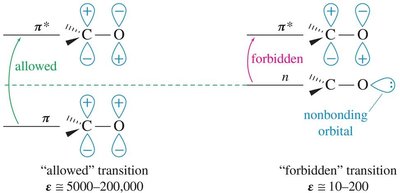
n → π* transitions are "forbidden" and have low molar absorptivity.
18.7 Review of Syntheses of Ketones and Aldehydes
Several methods exist for the synthesis of aldehydes and ketones:
Oxidation of alcohols
Friedel-Crafts acylation
Hydration and hydroboration-oxidation of alkynes
Ozonolysis of alkenes
Periodate cleavage of glycols
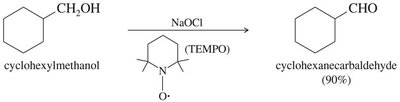
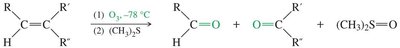
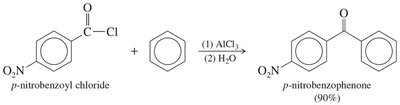
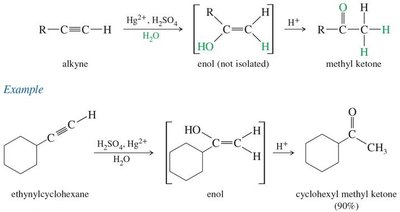
18.8 Synthesis of Ketones from Carboxylic Acids
Organolithium reagents can convert carboxylic acids to ketones via dianion intermediates, which are protonated and then lose water to yield the ketone.
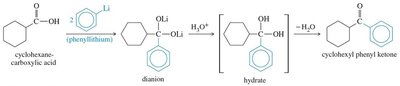
18.9 Synthesis of Ketones and Aldehydes from Nitriles
Grignard or organolithium reagents attack nitriles to form imines, which are hydrolyzed to ketones. Aluminum hydrides (e.g., DIBAL-H) can reduce nitriles to aldehydes.

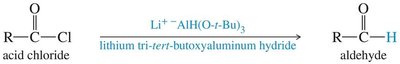
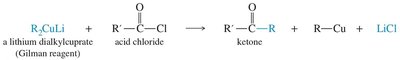
18.10 Synthesis from Acid Chlorides and Esters
Acid chlorides can be reduced to aldehydes or converted to ketones using lithium dialkyl cuprates (Gilman reagents). Grignard and organolithium reagents are not selective for this transformation.
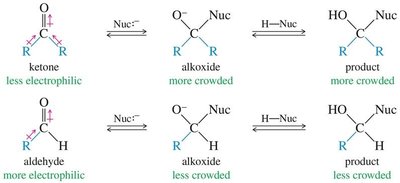
18.11 Reactions of Ketones and Aldehydes: Nucleophilic Addition
The most important reactions of aldehydes and ketones involve nucleophilic addition to the carbonyl carbon. Aldehydes are generally more reactive than ketones due to less steric hindrance and greater electrophilicity.

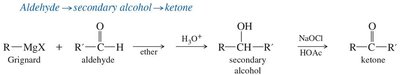
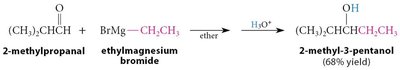
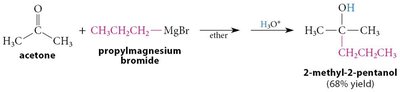
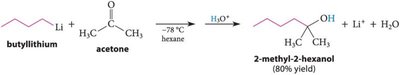
Grignard Addition: Grignard reagents add to carbonyls to form alcohols after protonation.
Organolithium and Acetylide Reagents: Similar to Grignard reagents, these form new C–C bonds.
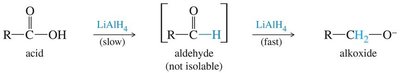
Reduction of Ketones and Aldehydes
Reduction of carbonyl compounds can be achieved using hydride sources such as sodium borohydride (NaBH4) and lithium aluminum hydride (LiAlH4).
NaBH4 is milder and more selective, reducing only aldehydes and ketones.
LiAlH4 is more reactive and can reduce carboxylic acids, esters, and amides as well.
Hydrogenation with a catalyst (e.g., Raney nickel) reduces both C=O and C=C bonds.
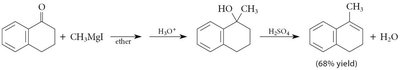
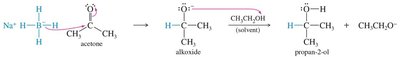
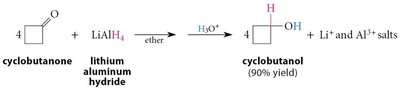

18.12 Hydration of Ketones and Aldehydes
In aqueous solution, aldehydes and ketones are in equilibrium with their hydrates (geminal diols). The equilibrium favors the unhydrated form for ketones. Hydration occurs via nucleophilic addition of water (acid-catalyzed) or hydroxide (base-catalyzed).
18.13 Formation of Cyanohydrins
Cyanohydrins are formed by nucleophilic addition of cyanide ion to the carbonyl group, followed by protonation. This reaction is base-catalyzed and is important for introducing a nitrile group into a molecule.
18.14 & 18.15 Formation of Imines and Related Condensations
Primary amines react with aldehydes or ketones to form imines (Schiff bases) via nucleophilic addition followed by dehydration. Hydroxylamine and hydrazines can also condense with carbonyls to form oximes and hydrazones, respectively.
18.16 Formation of Acetals
Acetals are formed by reaction of aldehydes or ketones with excess alcohol and acid catalyst. The mechanism proceeds via a hemiacetal intermediate. Acetal formation is reversible and is used to protect carbonyl groups during synthesis.
18.17 Use of Acetals as Protecting Groups
Acetals can be used as protecting groups for aldehydes and ketones, allowing selective reactions at other functional groups. They are stable in basic and neutral solutions but can be removed by acid and water.
18.18 The Wittig Reaction
The Wittig reaction is a key method for synthesizing alkenes from aldehydes and ketones using phosphorus ylides. The reaction proceeds via a concerted cycloaddition mechanism.
18.19 Oxidation of Aldehydes
Aldehydes are easily oxidized to carboxylic acids. The Tollens test is a classical method for detecting aldehydes, where silver ion is reduced to metallic silver upon oxidation of the aldehyde.
Class | General Formula |
|---|---|
Ketones | R–C(=O)–R' |
Aldehydes | R–C(=O)–H |
Carboxylic acids | R–C(=O)–OH |
Esters | R–C(=O)–O–R' |
Acid chlorides | R–C(=O)–Cl |
Amides | R–C(=O)–NH2 |
Additional info: This summary covers the structure, nomenclature, physical properties, spectroscopy, synthesis, and key reactions of aldehydes and ketones, as well as their use in organic synthesis. The included images reinforce structural, mechanistic, and spectroscopic concepts essential for mastery at the college organic chemistry level.
