 Back
BackAlkanes and Cycloalkanes: Properties, Reactions, and Mechanisms
Study Guide - Smart Notes

Alkanes and Cycloalkanes
Introduction to Organic Chemistry Reactions
Organic chemistry reactions are fundamental to understanding the behavior and transformation of organic molecules. These reactions can be classified by their mechanisms and energy profiles, which dictate their speed and spontaneity.
Addition (Combination, Synthesis, Direct Union): Two molecules combine to form a single product.
Elimination (Decomposition, Analysis): A single molecule splits into two products.
Single Displacement (Substitution): An atom or group is replaced by another.
Double Displacement (Metathesis, Exchange): Two compounds exchange parts.
Rearrangement: Structural reorganization, such as cis/trans interconversion.
Reaction Mechanisms
Organic reactions proceed via two main mechanisms: polar and radical. The mechanism influences the type of intermediates formed and the conditions required for the reaction.
Polar Mechanism: Involves heterolytic bond cleavage, forming ions. Common in reactions between nucleophiles (Nü) and electrophiles (e+).
Radical Mechanism: Involves homolytic bond cleavage, forming radicals (species with unpaired electrons). Typical for alkanes and cycloalkanes under heat (Δ) or light (hv).
Reaction Energy Diagrams and Transition States
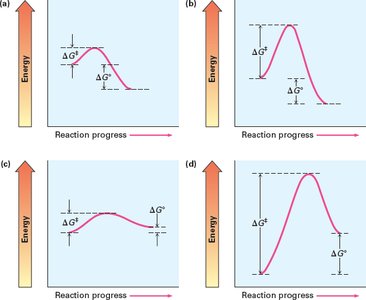
Energy diagrams illustrate the energy changes during a reaction, highlighting the activation energy and transition states. These diagrams help predict reaction speed and spontaneity.
Activation Energy (Ea): Minimum energy required to reach the transition state; lower Ea means faster reaction.
Transition State (Ts): High-energy state where reactants begin to form products.
Standard Free Energy Change (ΔG°): Indicates spontaneity; negative ΔG° is exergonic (energy-releasing), positive ΔG° is endergonic (energy-absorbing).
Classification of Reactions:
Exergonic (negative ΔG°): Fast or slow
Endergonic (positive ΔG°): Fast or slow
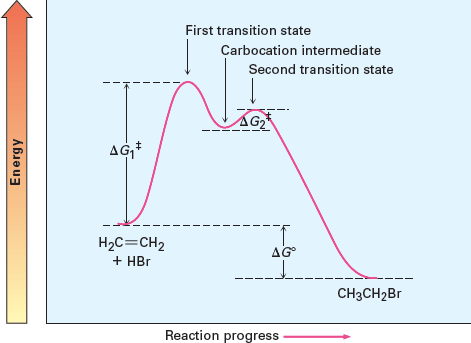
In multi-step reactions, intermediates such as carbocations may form, requiring additional activation energy for subsequent steps.
Alkanes and Cycloalkanes
Characteristics, Properties, and Types of Carbon Atoms
Alkanes and cycloalkanes are saturated hydrocarbons, meaning they contain only single bonds. Their physical and chemical properties are influenced by their structure and the types of carbon atoms present.
Alkanes: General formula ; found in petroleum and natural gas; exist as linear (normal) or branched chains.
Cycloalkanes: General formula ; cyclic structure with two fewer hydrogens than alkanes; can be substituted with various groups.

Physical Properties
Alkanes and cycloalkanes exhibit characteristic physical properties due to their non-polar nature.
Insoluble in water; exhibit London dispersion forces (non-polar intermolecular forces).
Lower density than water; float on water.
Low boiling points; increase with chain length due to more intermolecular forces.
Chemical Properties
Alkanes and cycloalkanes are generally unreactive but can undergo combustion and cracking.
Combustion: Reaction with O2 to produce CO2, H2O, heat, and light.
Cracking: Breaking large hydrocarbons into smaller ones using heat and/or Al2O3 catalyst.
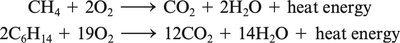
Combustion Equations:
Types of Carbon Atoms
Carbon atoms in alkanes and cycloalkanes are classified based on the number of other carbon atoms attached to them. This classification affects the stability of intermediates formed during reactions.
Type | Structure | Stability |
|---|---|---|
Primary (1°) | R-CH2-H | Least stable |
Secondary (2°) | R2-CH-H | More stable |
Tertiary (3°) | R3-C-H | Even more stable |
Quaternary (4°) | R4-C | Most stable |

Example: Methyl (1°), 2° carbon, 3° carbon, 4° carbon.
Radical Substitution Reactions (SR)
General Mechanism and Sample Reactions
Alkanes and cycloalkanes undergo radical substitution reactions, particularly halogenation, under specific conditions (heat or light). The mechanism involves three phases: initiation, propagation, and termination.
Initiation: Formation of radicals by homolytic cleavage of a halogen molecule (X2).
Propagation: Radicals react with alkanes, forming new radicals and products.
Termination: Combination of radicals to form stable molecules, ending the chain reaction.

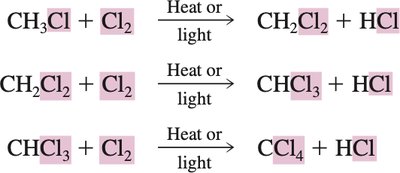
Example Reaction:

Multiple halogenations can occur, leading to mono-, di-, or poly-substituted products. Excess alkane is used to maximize monohalogenation.
Reactivity-Selectivity Principle
When multiple types of carbon atoms are present, the reactivity-selectivity principle helps predict which product will form. More reactive species are less selective, and the probability of product formation depends on both the number of available hydrogens and the reactivity of the radical.
Halogen | Relative Reactivity (3°) | Relative Reactivity (2°) | Relative Reactivity (1°) |
|---|---|---|---|
Cl• (RT) | 5.0x | 3.8x | 1.0x |
Br• (125°C) | 1600x | 82x | 1.0x |
Product Distribution in Halogenation
To determine the most likely product, calculate the expected yield based on the number of hydrogens and radical reactivity.
Chlorination of 2-methylpentane:
1°: 9 x 1.0 = 9 products (31%)
2°: 4 x 3.8 = 15 products (52%)
3°: 1 x 5.0 = 5 products (17%)
Total: 29 products

Most likely product: 2° alkyl chloride.
Bromination of 2-methylpentane:
1°: 9 x 1.0 = 9 products (0.4%)
2°: 4 x 82 = 328 products (17%)
3°: 1 x 1600 = 1600 products (82.6%)
Total: 1937 products

Most likely product: 3° alkyl bromide.
Summary Table: Radical Halogenation Product Distribution
Halogen | Most Likely Product | Reason |
|---|---|---|
Cl2 | 2° alkyl chloride | Moderate selectivity, moderate reactivity |
Br2 | 3° alkyl bromide | High selectivity, high reactivity |
Additional info: The notes cover Unit 4 of a pharmaceutical organic chemistry course, focusing on alkanes and cycloalkanes, their properties, and radical substitution reactions. The content is directly relevant to college-level organic chemistry, specifically chapters on alkanes, cycloalkanes, and radical reactions.
