 Back
BackAlkanes and Cycloalkanes: Structure, Nomenclature, Conformations, and Properties
Study Guide - Smart Notes

Alkanes and Cycloalkanes
Introduction to Hydrocarbons
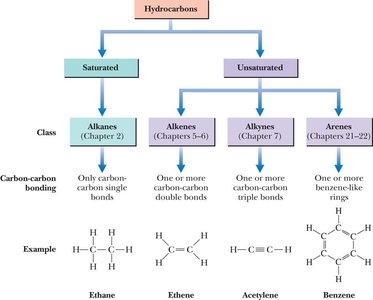
Hydrocarbons are organic molecules composed exclusively of carbon and hydrogen. They are classified based on the types of bonds between carbon atoms and the presence or absence of rings.
Saturated hydrocarbons: Only contain single bonds (alkanes and cycloalkanes).
Unsaturated hydrocarbons: Contain double or triple bonds, or aromatic rings (alkenes, alkynes, arenes).
Structure of Alkanes
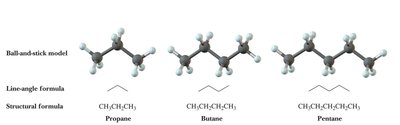
Alkanes are saturated hydrocarbons with only single bonds between carbon atoms. Each carbon is tetrahedral with bond angles of 109.5°.
General formula:
Line-angle formulas are commonly used for simplicity in organic chemistry.
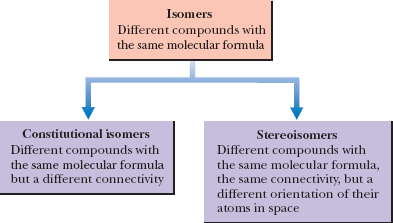
Constitutional Isomerism in Alkanes
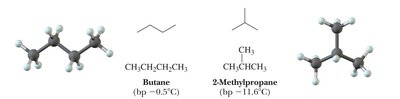
Constitutional isomers are compounds with the same molecular formula but different connectivity of atoms. This leads to different physical and chemical properties.
Example: C4H10 (butane and isobutane) are constitutional isomers.
Nomenclature of Alkanes and the IUPAC System
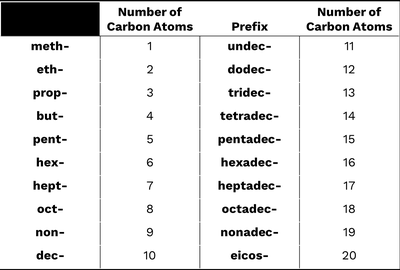
The IUPAC system provides a systematic way to name alkanes based on the number of carbons and the presence of substituents.
Find the longest continuous carbon chain (parent chain).
Name and number substituents to give the lowest possible numbers.
List substituents alphabetically; use prefixes (di-, tri-, etc.) for multiples of the same group.
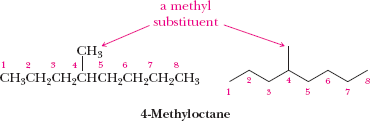
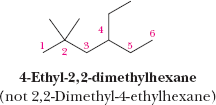
Alkyl Groups and Common Names
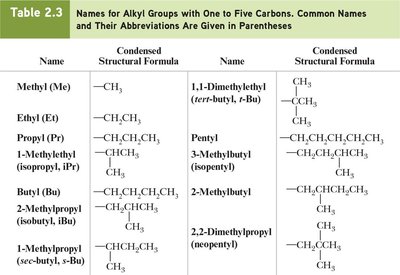
An alkyl group is derived from an alkane by removing one hydrogen atom. Common names are often used for simple alkyl groups and branched alkanes.
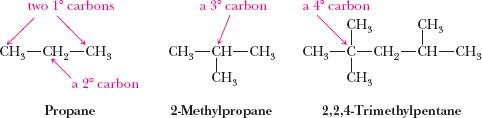
Classification of Carbon and Hydrogen Atoms
Carbons and hydrogens in alkanes are classified based on the number of other carbons to which they are attached:
Primary (1°): Attached to one other carbon
Secondary (2°): Attached to two other carbons
Tertiary (3°): Attached to three other carbons
Quaternary (4°): Attached to four other carbons
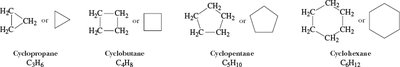
Cycloalkanes: Structure and Nomenclature
Cycloalkanes are saturated hydrocarbons with carbon atoms arranged in rings. The general formula is .
Named by adding the prefix "cyclo-" to the alkane name.
Number substituents to give the lowest set of numbers.
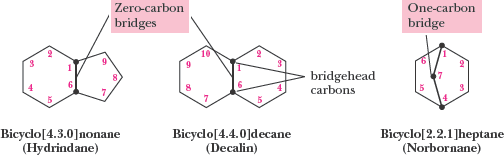
Bicycloalkanes
Bicycloalkanes contain two rings sharing two or more carbon atoms (bridgehead carbons). The general formula is .
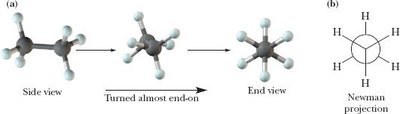
Conformations of Alkanes and Cycloalkanes
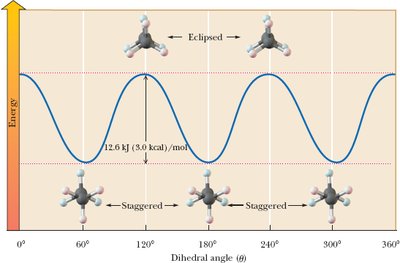
Conformations are different spatial arrangements of atoms resulting from rotation about single bonds. Strain in molecules can be torsional, steric, or angle strain.
Staggered conformation: Groups are as far apart as possible (lowest energy).
Eclipsed conformation: Groups are as close as possible (highest energy, torsional strain).
Newman projections are used to visualize conformations.

Cycloalkane Conformations
Cycloalkanes adopt non-planar conformations to minimize strain:
Cyclopropane: Planar, high angle and torsional strain.
Cyclobutane: Puckered to reduce torsional strain, but increases angle strain.
Cyclopentane: Envelope conformation to relieve torsional strain.
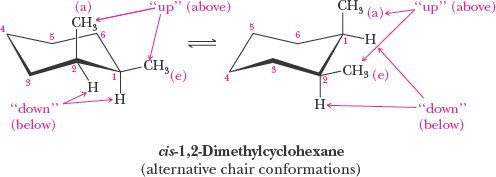
Cyclohexane: Chair conformation is most stable; all bonds are staggered and angles are near 109.5°.

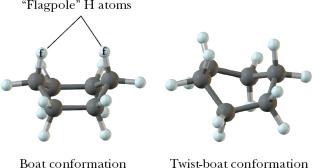
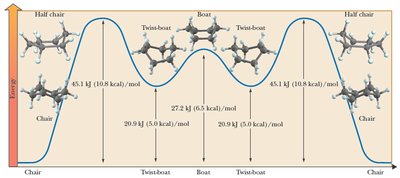
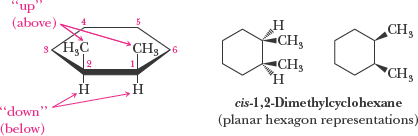
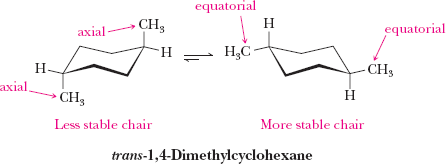
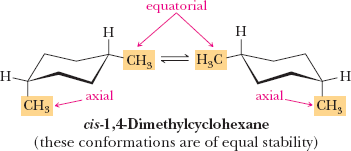
Axial and Equatorial Positions in Cyclohexane
In the chair conformation, hydrogens (or substituents) can be axial (parallel to the ring axis) or equatorial (around the ring's equator). Substituents prefer the equatorial position to minimize steric strain (diaxial interactions).
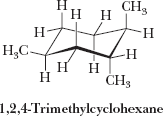
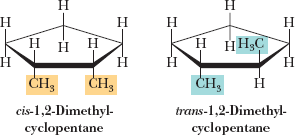
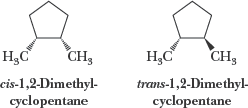
Cis-Trans Isomerism in Cycloalkanes
Cycloalkanes with two or more substituents can exhibit cis-trans (geometric) isomerism, depending on whether the substituents are on the same or opposite sides of the ring.
Cis: Substituents on the same side
Trans: Substituents on opposite sides
These isomers cannot interconvert without breaking bonds.
Physical Properties of Alkanes and Cycloalkanes
Alkanes and cycloalkanes are nonpolar and interact via weak dispersion forces. Their physical properties depend on molecular weight and branching.
Boiling points increase with molecular weight and decrease with branching.
Melting points increase less regularly with molecular weight.
Low molecular weight alkanes are gases; higher ones are liquids or solids at room temperature.
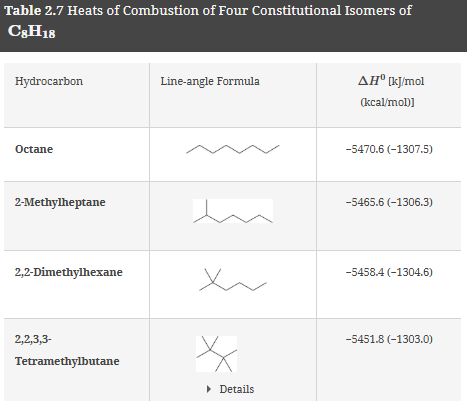
Heats of Combustion and Stability
Combustion of alkanes produces CO2 and H2O, releasing energy (heat of combustion). More branched alkanes are more stable and have lower heats of combustion per CH2 group. Cycloalkanes with significant ring strain (e.g., cyclopropane) release more energy upon combustion.
Example equations:
Methane combustion:
Propane combustion:
Key concept: The more branched the alkane, the more stable it is, and the lower its heat of combustion.
