 Back
BackAlkanes, Cycloalkanes, and Functional Groups: Structure, Properties, and Nomenclature
Study Guide - Smart Notes

Functional Groups in Organic Chemistry
Definition and Importance of Functional Groups
Functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. The presence and identity of a functional group largely determine the reactivity and properties of organic compounds.
Definition: A functional group is a group of atoms within a molecule that has a characteristic chemical behavior.
Consistency: Functional groups behave similarly in different molecules, regardless of the molecule's size or complexity.
Example: Both ethylene and menthene contain a carbon–carbon double bond and react with bromine in the same way, adding Br atoms across the double bond.
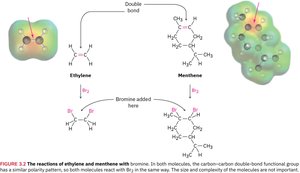
Additional info: Functional groups are the foundation for classifying organic compounds and predicting their reactivity.
Common Functional Groups and Their Structures
Organic compounds are classified based on the functional groups they contain. Some common functional groups include alkenes, alkynes, alcohols, ethers, amines, and carbonyl compounds.
Functional Group | Structure | Example |
|---|---|---|
Alkene | –C=C– | Ethylene |
Alkyne | –C≡C– | Acetylene |
Alcohol | –OH | Ethanol |
Ether | –O– | Dimethyl ether |
Aldehyde | –CHO | Formaldehyde |
Ketone | –CO– | Acetone |
Carboxylic acid | –COOH | Acetic acid |
Amine | –NH2 | Methylamine |


Classification by Bond Types
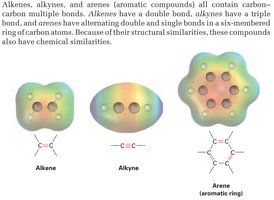
Carbon–Carbon Multiple Bonds: Alkenes (double bonds), alkynes (triple bonds), and arenes (aromatic rings) all contain carbon–carbon multiple bonds, which impart unique chemical properties.
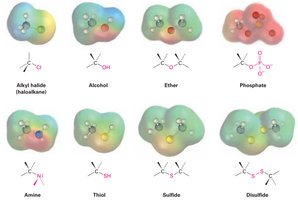
Carbon Singly Bonded to Electronegative Atoms: Alkyl halides, alcohols, ethers, amines, thiols, and related groups feature a carbon atom singly bonded to halogen, oxygen, nitrogen, or sulfur.
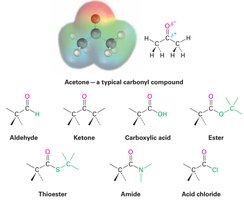
Carbonyl Groups (C=O): Carbonyl groups are present in aldehydes, ketones, carboxylic acids, esters, amides, and related compounds. The carbonyl carbon is electrophilic (δ+), and the oxygen is nucleophilic (δ−).
Alkanes: Structure, Types, and Properties
Definition and General Formula
Alkanes are saturated hydrocarbons, meaning they contain only single bonds between carbon atoms. Their general formula is , where n is the number of carbon atoms.
Saturated Hydrocarbons: Each carbon atom is bonded to as many hydrogen atoms as possible.
Aliphatic Compounds: Alkanes are sometimes called aliphatic compounds, derived from the Greek word for "fat," due to their presence in animal fats.
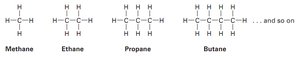
Types of Alkanes
Alkanes can be classified based on their carbon skeletons:
Straight-chain alkanes: Carbon atoms are connected in a single, unbranched chain.
Branched-chain alkanes: The carbon chain contains one or more branches.
Cycloalkanes: The carbon atoms form a ring structure.
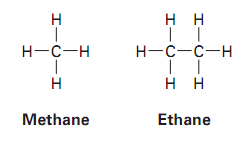
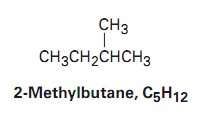
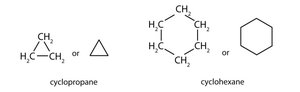
Isomerism in Alkanes
Isomers are compounds with the same molecular formula but different arrangements of atoms. Constitutional isomers differ in the connectivity of their atoms.
Constitutional Isomers: Same formula, different connectivity and properties.
Example: Butane and isobutane (C4H10) are constitutional isomers.
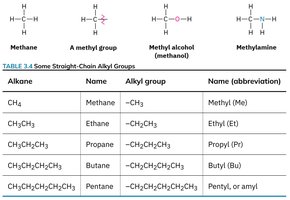
Alkyl Groups
An alkyl group is formed by removing one hydrogen atom from an alkane. Alkyl groups are often represented as R– and are important as substituents in larger molecules.
Examples: Methyl (–CH3), ethyl (–C2H5), propyl (–C3H7).
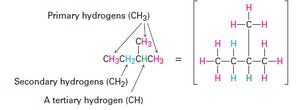
Classification of Carbon and Hydrogen Atoms
Carbons and hydrogens in alkanes are classified based on the number of other carbons to which they are attached:
Primary (1°): Bonded to one other carbon
Secondary (2°): Bonded to two other carbons
Tertiary (3°): Bonded to three other carbons
Quaternary (4°): Bonded to four other carbons

Nomenclature of Alkanes (IUPAC System)
Principles of IUPAC Nomenclature
The IUPAC system provides a systematic method for naming organic compounds. The name is constructed from four parts: locant, prefix, parent, and suffix.
Locant: Position of substituents and functional groups
Prefix: Identity of substituents
Parent: Number of carbons in the main chain
Suffix: Primary functional group

Steps in Naming Alkanes
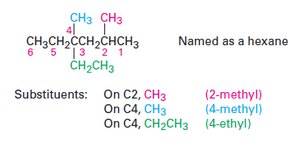
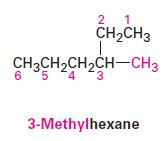
Identify the parent hydrocarbon: Find the longest continuous carbon chain.
Number the chain: Assign numbers to the carbon atoms to give the substituents the lowest possible numbers.
Identify and number substituents: Name and locate each substituent.
Write the name: Combine the parts into a single word, listing substituents alphabetically.
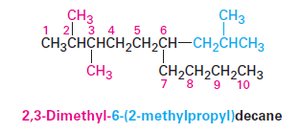
Name complex substituents: Name as though they are themselves compounds.

Physical Properties and Reactions of Alkanes
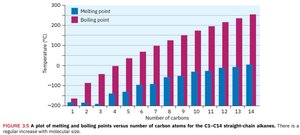
Boiling and Melting Points
Alkanes exhibit regular increases in boiling and melting points as molecular weight increases. This is due to the increase in dispersion forces (London forces) between larger molecules.
Chemical Reactions of Alkanes
Combustion: Alkanes react with oxygen to produce carbon dioxide, water, and energy.
Halogenation: Alkanes react with halogens (e.g., Cl2) under UV light to form alkyl halides and hydrogen halide.
Example combustion reaction:

Conformations and Stereochemistry of Alkanes
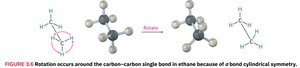
Bond Rotation and Conformations
Stereochemistry studies the three-dimensional arrangement of atoms in molecules. In alkanes, rotation around C–C single bonds leads to different spatial arrangements called conformations.
Conformers: Different spatial arrangements due to rotation around single bonds.
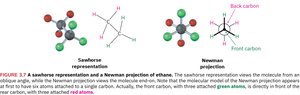
Sawhorse and Newman Projections: Two common ways to represent conformations. Sawhorse shows the molecule at an angle; Newman projection looks straight down the bond axis.
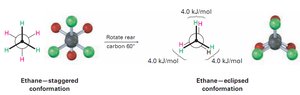
Staggered and Eclipsed Conformations
The most stable conformation of ethane is the staggered form, where all C–H bonds are as far apart as possible. The least stable is the eclipsed form, where C–H bonds are aligned.
Staggered: Lower energy, more stable
Eclipsed: Higher energy, less stable
Cycloalkanes: Structure and Conformations
Definition and Properties
Cycloalkanes are saturated hydrocarbons with carbon atoms arranged in a ring. Their general formula is .
Less Flexible: Cycloalkanes are less flexible than straight-chain alkanes due to restricted rotation around ring bonds.

Chair Conformation of Cyclohexane
Cyclohexane adopts a strain-free, three-dimensional chair conformation, which minimizes torsional and angle strain.
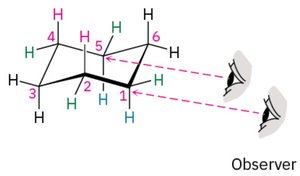
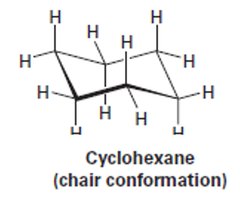
Drawing the Chair Conformation
The chair conformation can be drawn in three steps: drawing two parallel lines, connecting the topmost and bottommost carbons, and completing the bonds.

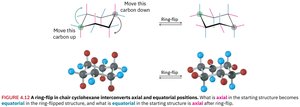
Axial and Equatorial Positions
In the chair conformation, each carbon has one axial (parallel to the ring axis) and one equatorial (around the ring equator) substituent position.
Ring Flip in Cyclohexane
A ring flip interconverts axial and equatorial positions, allowing the molecule to relieve steric strain. This process is rapid at room temperature.
Additional info: Understanding the structure, nomenclature, and conformational analysis of alkanes and cycloalkanes is foundational for further study in organic chemistry, including reactions, mechanisms, and the behavior of more complex molecules.
