 Back
BackAlkanes, Cycloalkanes, Isomerism, and Stereochemistry: Step-by-Step Organic Chemistry Guidance
Study Guide - Smart Notes

Q1. Do the condensed formulas in each pair represent the same compound or constitutional isomers?
Background
Topic: Constitutional Isomerism in Alkanes
This question tests your ability to distinguish between compounds with the same molecular formula but different connectivity (constitutional isomers) versus those that are simply different representations of the same molecule.
Key Terms and Concepts:
Constitutional isomers: Compounds with the same molecular formula but different connectivity of atoms.
Condensed formula: A way of writing organic molecules to show how atoms are connected without drawing every bond.
Step-by-Step Guidance
For each pair, write out the full structural formula from the condensed formula to visualize the connectivity of the carbon atoms.
Compare the connectivity of the carbon chains and the position of any branches or substituents.
Check if the two structures can be superimposed (i.e., are the same compound) or if they have different connections (making them constitutional isomers).
Remember, even if the molecular formula is the same, a different arrangement of atoms means they are constitutional isomers.
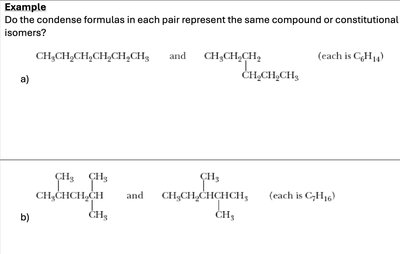
Try solving on your own before revealing the answer!
Q2. Write line-angle formulas for the five constitutional isomers with the molecular formula C6H14.
Background
Topic: Drawing Isomers of Alkanes
This question tests your ability to draw all possible constitutional isomers for a given alkane formula using line-angle (skeletal) notation.
Key Terms and Concepts:
Line-angle formula: A simplified organic structure where lines represent bonds and vertices represent carbon atoms.
Isomer: Compounds with the same molecular formula but different structures.
Step-by-Step Guidance
Recall that C6H14 is the formula for hexane and its isomers.
Start by drawing the straight-chain (n-hexane) structure.
Systematically move one or more methyl groups to create branched isomers, ensuring each structure is unique and not a duplicate by rotation or reflection.
Check that each structure has six carbons and fourteen hydrogens.

Try solving on your own before revealing the answer!
Q3. Write the IUPAC and common names for these alkanes.
Background
Topic: Alkane Nomenclature
This question tests your ability to apply IUPAC rules and recognize common names for branched alkanes.
Key Terms and Concepts:
IUPAC name: The systematic name based on the longest carbon chain and substituent positions.
Common name: Traditional names, often used for simple branched alkanes.
Step-by-Step Guidance
Identify the longest continuous carbon chain in each structure (this gives the parent name).
Number the chain from the end nearest a substituent to assign the lowest possible numbers to the branches.
Name and number each substituent, listing them in alphabetical order for the IUPAC name.
For the common name, recognize if the structure matches a known alkane (e.g., isopropyl, tert-butyl, etc.).
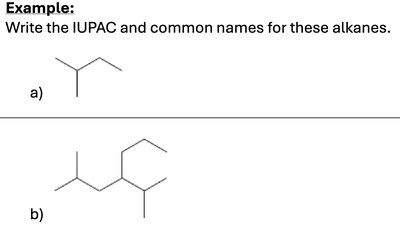
Try solving on your own before revealing the answer!
Q4. Write the molecular formula and the IUPAC name for each cycloalkane.
Background
Topic: Cycloalkane Nomenclature and Formulas
This question tests your ability to name cycloalkanes and determine their molecular formulas.
Key Terms and Concepts:
Cycloalkane: A saturated hydrocarbon with carbon atoms arranged in a ring.
Molecular formula: Shows the number of each type of atom in a molecule.
IUPAC naming: Identify the ring size and name substituents with appropriate locants.
Step-by-Step Guidance
Count the number of carbons in the ring and any substituents to determine the molecular formula.
Name the parent ring (e.g., cyclopentane, cyclohexane, etc.).
Identify and number substituents to give the lowest possible numbers, following IUPAC rules.
Combine the substituent names and locants with the parent ring name.
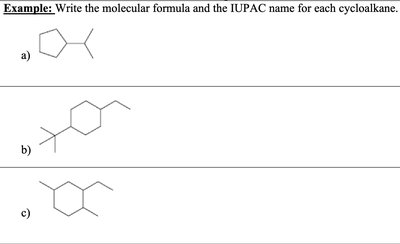
Try solving on your own before revealing the answer!
Q5. Write molecular formulas for each bicycloalkane, given its number of carbon atoms.
Background
Topic: Bicycloalkane Nomenclature and Formulas
This question tests your understanding of how to determine the molecular formula for bicyclic alkanes based on their carbon count.
Key Terms and Concepts:
Bicycloalkane: A saturated hydrocarbon with two rings sharing two or more carbons.
General formula: for bicycloalkanes.
Step-by-Step Guidance
Identify the number of carbon atoms in each bicycloalkane (given in the question).
Apply the general formula for bicycloalkanes: , where is the number of carbons.
Substitute the value of for each compound to find the molecular formula.
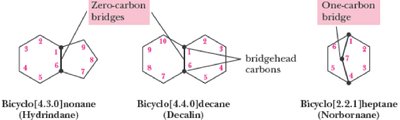

Try solving on your own before revealing the answer!
