Back
BackAlkenes: Structure, Reactivity, and Key Reactions
Study Guide - Smart Notes

Alkenes: Structure and Reactivity
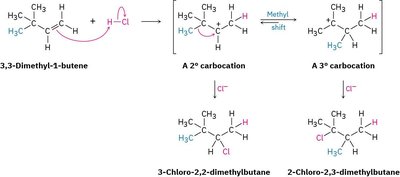
Carbocation Rearrangements – Methyl Shift
Carbocation rearrangements are important in the chemistry of alkenes, especially during reactions that proceed via carbocation intermediates. A common rearrangement is the methyl shift, where a methyl group migrates to stabilize the carbocation, often converting a less stable secondary carbocation into a more stable tertiary carbocation.
Carbocation Stability: Tertiary carbocations are more stable than secondary or primary due to hyperconjugation and inductive effects.
Methyl Shift: Occurs when a methyl group adjacent to a carbocation migrates, resulting in a more stable carbocation intermediate.
Example: In the reaction of 3,3-dimethyl-1-butene with HCl, a methyl shift leads to the formation of a tertiary carbocation, which then reacts with Cl- to give the major product.
Alkenes: Reactions and Synthesis
Overview of Alkene Reactions
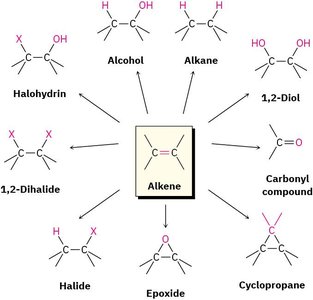
Alkenes are versatile intermediates in organic synthesis, undergoing a variety of addition and elimination reactions. The double bond acts as a nucleophile, reacting with electrophiles to form a wide range of products.
Types of Reactions: Alkenes can be converted into alcohols, halohydrins, 1,2-diols, carbonyl compounds, halides, epoxides, cyclopropanes, and more.
Applications: These transformations are foundational in the synthesis of complex organic molecules.
Preparing Alkenes: Elimination Reactions
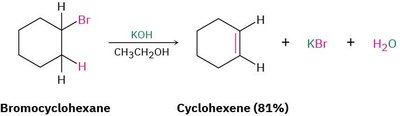
Alkenes are commonly prepared by elimination reactions, which involve the removal of atoms or groups from adjacent carbons in a molecule, resulting in the formation of a double bond.
Dehydrohalogenation: The elimination of HX (where X is a halide) from an alkyl halide using a strong base produces an alkene.
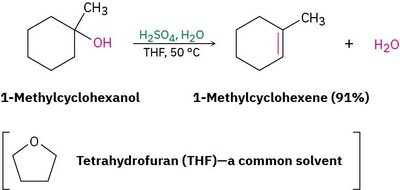
Dehydration: The elimination of water from an alcohol, typically using a strong acid, yields an alkene.
Example: Bromocyclohexane treated with KOH in ethanol gives cyclohexene.
Example: 1-Methylcyclohexanol undergoes acid-catalyzed dehydration to form 1-methylcyclohexene.
Halogenation of Alkenes: Addition of X2
Mechanism and Stereochemistry
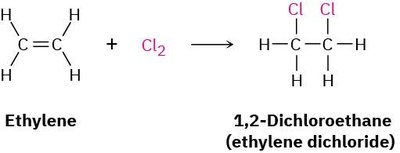
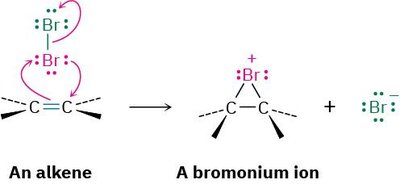
Halogenation is the addition of halogens (Br2 or Cl2) to alkenes, resulting in 1,2-dihalides. The reaction is stereospecific and typically proceeds via anti addition, meaning the two halogen atoms add to opposite faces of the double bond.
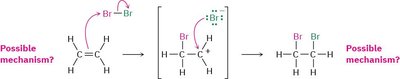
Mechanism: The alkene reacts with the halogen molecule to form a cyclic halonium ion intermediate, which is then attacked by a halide ion from the opposite side.
Anti Stereochemistry: The product is a trans-1,2-dihalide due to the anti addition mechanism.
Example: Ethylene reacts with Cl2 to form 1,2-dichloroethane.
Possible Mechanism: The reaction proceeds through a three-membered halonium ion intermediate rather than a carbocation.
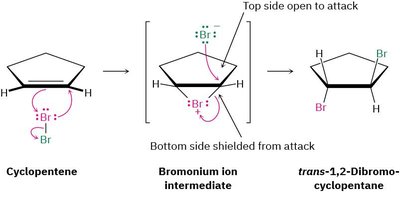
Example: Cyclopentene reacts with Br2 to give only the trans-1,2-dibromocyclopentane, not the cis isomer.
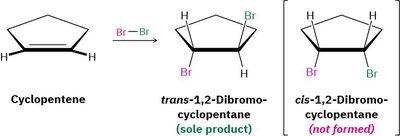
Bromonium Ion Intermediate: The formation of a bromonium ion explains the observed anti stereochemistry.
Mechanistic Details: The bromonium ion shields one face of the alkene, so the nucleophile attacks from the opposite side, leading to anti addition.
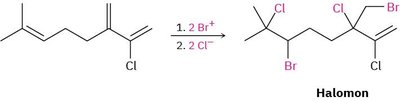
Halogenation in Complex Systems
Example: Halomon is formed by the addition of Br+ and Cl- to a polyene system.
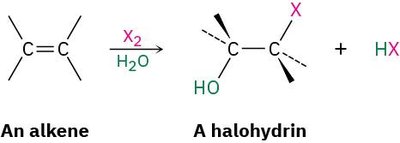
Halohydrin Formation: Addition of HO-X
Halohydrins are formed when alkenes react with halogens in the presence of water. The product is a 1,2-halo alcohol, with the halogen and hydroxyl group on adjacent carbons.
Mechanism: The alkene first forms a halonium ion, which is then attacked by water (a nucleophile), leading to anti addition of the halogen and hydroxyl group.
Example: An alkene reacts with X2 and H2O to form a halohydrin and HX.
Stepwise Mechanism: 1) Formation of bromonium ion, 2) Nucleophilic attack by water, 3) Deprotonation to yield the halohydrin.
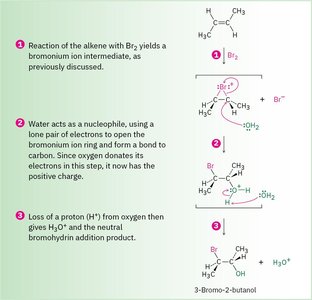
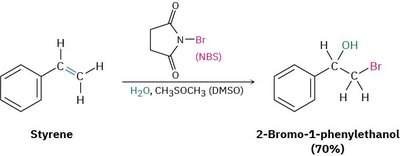
Practical Considerations: Since many alkenes are not water-soluble, halohydrin formation is often performed in aqueous DMSO using N-bromosuccinimide (NBS) as the bromine source.
Example: Styrene reacts with NBS in aqueous DMSO to give 2-bromo-1-phenylethanol.
Biological Example: Halohydrin formation can also be catalyzed by haloperoxidase enzymes in the presence of H2O2 and Br-.
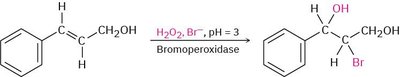
Hydration of Alkenes: Addition of H2O
Acid-Catalyzed Hydration
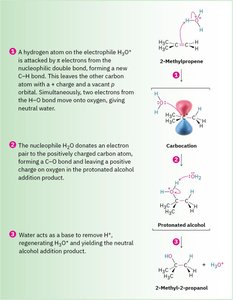
Hydration is the addition of water to an alkene to form an alcohol. This reaction is typically catalyzed by a strong acid and follows Markovnikov's rule, where the proton adds to the carbon with more hydrogens, and the hydroxyl group adds to the more substituted carbon.
Mechanism: 1) Protonation of the alkene to form a carbocation, 2) Nucleophilic attack by water, 3) Deprotonation to yield the alcohol.
Example: Ethylene reacts with water in the presence of H3PO4 catalyst to form ethanol.
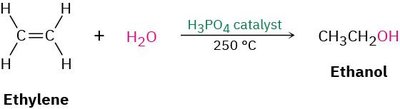
Detailed Mechanism: Illustrated for 2-methylpropene, showing carbocation formation, nucleophilic attack, and deprotonation.
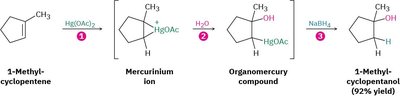
Oxymercuration–Demercuration
Oxymercuration–demercuration is a two-step method for hydrating alkenes that avoids carbocation rearrangements. The reaction proceeds via electrophilic addition of Hg2+ to the alkene, followed by reduction with NaBH4 to yield the alcohol.
Markovnikov Addition: The hydroxyl group attaches to the more substituted carbon, following Markovnikov's rule.
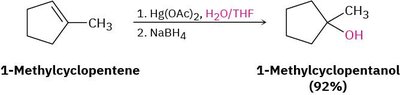
Example: 1-Methylcyclopentene is converted to 1-methylcyclopentanol using Hg(OAc)2, H2O/THF, and NaBH4.
Mechanistic Analogy: The mechanism is closely related to halohydrin formation, involving a mercurinium ion intermediate.
Stepwise Mechanism: 1) Formation of mercurinium ion, 2) Nucleophilic attack by water, 3) Reduction by NaBH4 to yield the alcohol.