 Back
BackAlkyl Halides and Alcohols: Nucleophilic Substitution and Related Concepts
Study Guide - Smart Notes

Alkyl Halides and Alcohols: Structure, Properties, and Nucleophilic Substitution
Introduction to Alkyl Halides and Alcohols
Alkyl halides and alcohols are fundamental classes of organic compounds, each containing a functional group that defines their chemical reactivity. Understanding their structure, nomenclature, and reactivity is essential for mastering organic chemistry mechanisms, especially nucleophilic substitution reactions.
Alkyl Halides (R-X): Compounds where a halogen (X = Cl, Br, I, F) is bonded to an sp3 hybridized carbon atom.
Alcohols (R-OH): Compounds where a hydroxyl group (-OH) is bonded to an sp3 hybridized carbon atom.
Functional Group: The atom or group in a molecule most responsible for its characteristic reactions.
Mechanism: The stepwise process by which reactants are converted to products.

Classification of Halogenated Organic Compounds
Types of Halides
Alkyl Halides: Halogen attached to an sp3 carbon of an alkyl group.
Vinyl Halides: Halogen attached to an sp2 carbon of an alkene.
Aryl Halides: Halogen attached to an aromatic ring.
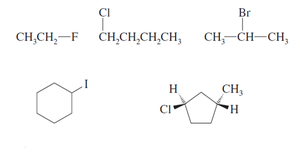
Geminal Dihalide: Two halogens on the same carbon. Vicinal Dihalide: Two halogens on adjacent carbons.
Bonding and Structure
Hybridization and Bond Angles
In both alcohols and alkyl halides, the carbon bearing the functional group is sp3 hybridized, resulting in tetrahedral bond angles (~109.5°).
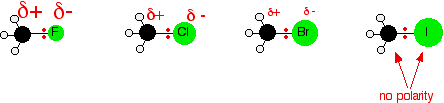
Bond Lengths: In alkyl halides, the C–X bond length increases as the halogen increases in size: C–F < C–Cl < C–Br < C–I.
Polarity: Both classes are polar molecules with significant dipole moments due to the electronegativity difference between carbon and the attached group.
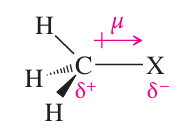
Physical Properties
Boiling Points and Intermolecular Forces
Alcohols generally have much higher boiling points than alkyl halides due to hydrogen bonding. Alkyl halides rely mainly on London dispersion forces, which increase with molecular size and polarizability.
Alcohols: Exhibit hydrogen bonding, leading to higher boiling points.
Alkyl Halides: Boiling points increase with the size and polarizability of the halogen, except for fluorine, which has low polarizability.
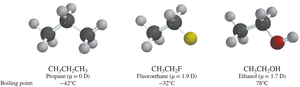
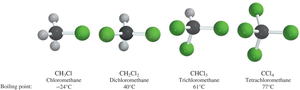
Polarizability
Polarizability refers to how easily the electron cloud of a molecule can be distorted. Larger, more diffuse electron clouds (as in heavier halogens) are more polarizable, leading to stronger London forces and higher boiling points. Fluorine, being small and tightly held, is much less polarizable.
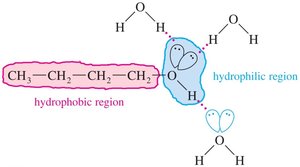
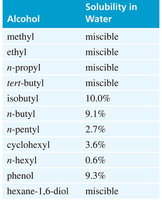
Solubility in Water
Alcohols and alkyl halides differ significantly in water solubility:
Alcohols: Low molecular weight alcohols are miscible with water due to hydrogen bonding. As the alkyl chain length increases, solubility decreases due to the hydrophobic effect.
Alkyl Halides: Generally insoluble in water.
Density
Alkyl fluorides and chlorides are less dense than water; bromides and iodides are denser.
Polyhalogenation increases density (e.g., CH2Cl2, CHCl3, CCl4).
All liquid alcohols are less dense than water (~0.8 g/mL).
Nomenclature
IUPAC Nomenclature of Alkyl Halides
Functional Class Nomenclature: Name the alkyl group and the halide as separate words (e.g., ethyl chloride).
Substitutive Nomenclature: Treat the halogen as a substituent (fluoro-, chloro-, bromo-, iodo-) on the parent alkane chain. Number the chain to give the substituent the lowest possible number.
IUPAC Nomenclature of Alcohols
Functional Class Names: Name the alkyl group bearing the -OH, followed by 'alcohol' (e.g., isopropyl alcohol).
Substitutive Names: Identify the longest chain with the -OH group, replace the -e ending with -ol, and number the chain to give the -OH the lowest possible number.
For multiple -OH groups, use di-, tri-, etc. (e.g., 2,3-hexanediol).
Classification of Alcohols and Alkyl Halides
Primary, Secondary, and Tertiary
Primary (1°): Carbon with the functional group is bonded to one other carbon.
Secondary (2°): Carbon with the functional group is bonded to two other carbons.
Tertiary (3°): Carbon with the functional group is bonded to three other carbons.
Aromatic (Phenol): -OH group bonded to a benzene ring.
Carbocations: Structure, Stability, and Rearrangements
Carbocation Stability
Carbocations are intermediates in many organic reactions. Their stability increases with the number of alkyl groups attached to the positively charged carbon due to hyperconjugation and inductive effects:
Methyl < Primary < Secondary < Tertiary
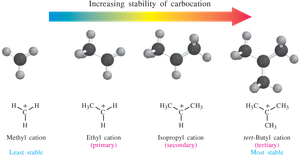
Carbocation Rearrangements
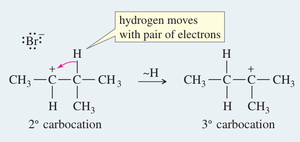
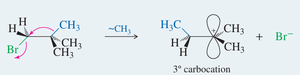
Carbocations can rearrange via hydride or alkyl shifts to form more stable carbocations. These rearrangements are common in reactions proceeding through carbocation intermediates (e.g., SN1 reactions).
Hydride Shift: Migration of a hydrogen atom with its electron pair to an adjacent carbocation.
Methyl Shift: Migration of a methyl group with its electron pair to an adjacent carbocation.
Types of Organic Reactions
Overview
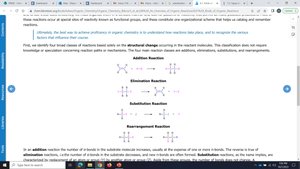
Addition: Increase in the number of σ-bonds, usually at the expense of π-bonds.
Elimination: Decrease in the number of σ-bonds, formation of new π-bonds.
Substitution: Replacement of one atom or group by another.
Rearrangement: Formation of an isomer by migration of atoms or groups within the molecule.
Nucleophiles and Electrophiles
Definitions
Nucleophile: Electron-rich species that donates a pair of electrons to form a new covalent bond ("nucleus-loving").
Electrophile: Electron-deficient species that accepts a pair of electrons ("electron-loving").
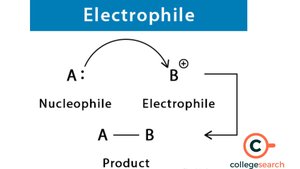
SN1 and SN2 Reaction Mechanisms
SN1 Reaction (Substitution Nucleophilic Unimolecular)
The SN1 reaction proceeds via a two-step mechanism and is first-order with respect to the substrate. It involves the formation of a carbocation intermediate and is favored by tertiary substrates and polar protic solvents.
Step 1: Leaving group departs, forming a carbocation (rate-determining step).
Step 2: Nucleophile attacks the carbocation.
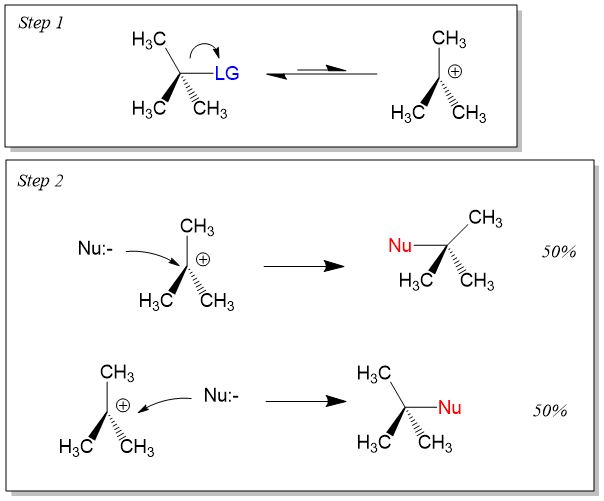
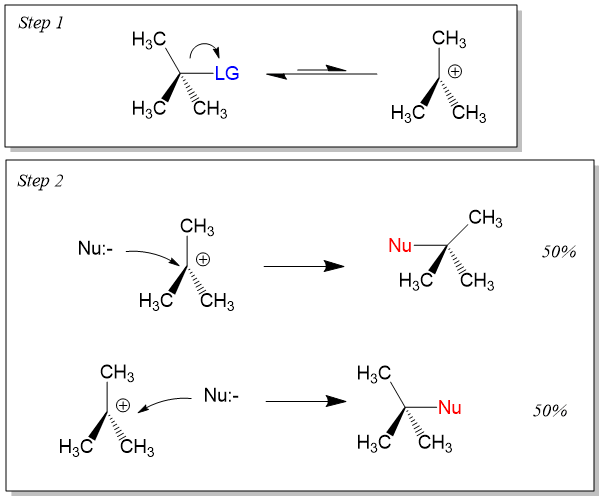
Stereochemistry of SN1
Because the carbocation intermediate is planar, the nucleophile can attack from either side, leading to racemization or partial inversion of configuration.
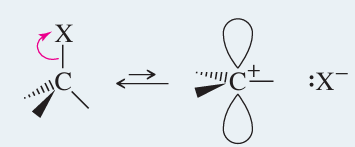
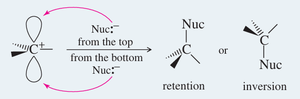
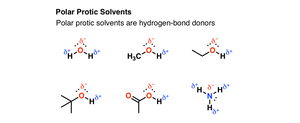
Solvent Effects in SN1
SN1 reactions are faster in polar protic solvents (e.g., water, alcohols) because these solvents stabilize the carbocation and the leaving group through solvation and hydrogen bonding.
SN2 Reaction (Substitution Nucleophilic Bimolecular)
The SN2 reaction is a one-step, concerted mechanism where the nucleophile attacks the substrate from the opposite side of the leaving group, resulting in inversion of configuration. It is second-order overall and favored by primary substrates and polar aprotic solvents.
Mechanism: Nucleophile attacks as the leaving group departs; both events occur simultaneously.
Stereochemistry: Always results in inversion at the stereocenter (Walden inversion).
Factors Affecting SN2 Reactions
Nucleophile Strength: Stronger nucleophiles increase the rate.
Steric Hindrance: Bulky substrates or nucleophiles slow the reaction.
Solvent: Polar aprotic solvents (e.g., DMSO, acetone) enhance nucleophilicity and reaction rate.
Leaving Group: Good leaving groups are weak bases and highly polarizable (e.g., I- > Br- > Cl- > F-).
Comparison of SN1 and SN2 Mechanisms
Feature | SN1 | SN2 |
|---|---|---|
Order | First order (substrate only) | Second order (substrate + nucleophile) |
Mechanism | Two-step (carbocation intermediate) | One-step (concerted) |
Stereochemistry | Racemization/partial inversion | Inversion only |
Substrate Preference | Tertiary > Secondary > Primary | Primary > Secondary > Tertiary |
Solvent | Polar protic | Polar aprotic |
Carbocation Rearrangement | Possible | Not possible |
Preparation of Alkyl Halides from Alcohols
Reaction with Hydrogen Halides
Alcohols react with hydrogen halides (HX) to form alkyl halides and water. The reactivity order of hydrogen halides is HI > HBr > HCl > HF. Tertiary alcohols react fastest, primary slowest.
Mechanism: Tertiary alcohols react via SN1; primary via SN2.
Other Methods
Thionyl Chloride (SOCl2): Converts alcohols to alkyl chlorides, often in the presence of a base like pyridine.
Phosphorus Halides (PCl3, PBr3): Convert alcohols to alkyl halides.
Practice Problems and Applications
Apply IUPAC nomenclature rules to name complex alkyl halides and alcohols.
Predict the product and mechanism (SN1 or SN2) for the reaction of a given alcohol with a hydrogen halide.
Classify alcohols and alkyl halides as primary, secondary, or tertiary based on their structure.
Identify the most stable carbocation in a set of structures.
Explain the effect of solvent, nucleophile, and leaving group on the rate and outcome of nucleophilic substitution reactions.
Additional info: This guide integrates textbook content, lecture notes, and visual aids to provide a comprehensive overview of nucleophilic substitution in alkyl halides and alcohols, suitable for exam preparation and foundational understanding in organic chemistry.
