 Back
BackAlpha Carbon Chemistry and Enolate Reactions in Organic Synthesis
Study Guide - Smart Notes

Alpha Carbon Chemistry
Introduction to Alpha Carbon Chemistry
The chemistry of the alpha carbon— the carbon atom directly adjacent to a carbonyl group— is central to many important organic reactions. The acidity of alpha hydrogens, the formation of enolates, and their subsequent reactions enable the construction of complex carbon frameworks in both laboratory and biological settings.
Keto-Enol Tautomerism
Keto-Enol Equilibrium
Keto-enol tautomerism is the equilibrium between a ketone (or aldehyde) and its corresponding enol form. This equilibrium is typically shifted toward the keto form, but can be influenced by structural factors and solvent effects.
Keto form: The standard carbonyl structure (C=O).
Enol form: Contains a C=C double bond and an alcohol (OH) group.
Equilibration: Accelerated by trace acid or base.
Solvent effects: Hydrogen bonding in water can stabilize the enol form, but H-bonds are relatively weak in water compared to other solvents.
Example: Acetone exists predominantly in the keto form, but the enol content can increase with certain substituents or under specific conditions.
Acidity of Alpha Hydrogens
pKa Values and Factors Affecting Acidity
The acidity of alpha hydrogens is crucial for enolate formation. Electron-withdrawing groups (such as -CN, -SO2R, -NO2) increase acidity by stabilizing the resulting anion.
pKa values: Lower pKa indicates higher acidity. Typical pKa values for alpha hydrogens in ketones and esters range from 9 to 25.
Resonance stabilization: Resonance in the anion greatly increases acidity, especially in aromatic systems (e.g., para phenol is more acidic than phenol).
Solvent effects: Most pKa values are measured in water, but organic reactions often use other solvents.
Enolate Formation and Reactivity
Enolate Generation
Enolates are generated by deprotonating the alpha carbon of a carbonyl compound using a strong base. The choice of base and reaction conditions determines whether the kinetic or thermodynamic enolate is formed.
Kinetic enolate: Formed rapidly, less substituted, favored by strong, bulky bases (e.g., LDA) at low temperatures.
Thermodynamic enolate: More substituted, favored by weaker bases at higher temperatures.
Halogenation of Carbonyl Compounds
Alpha Halogenation
Ketones and aldehydes can undergo alpha halogenation with halogens (Cl2, Br2, I2) in acidic or basic conditions. The reaction often exhibits autocatalysis, where the product accelerates the reaction.
Autocatalytic behavior: The reaction starts slowly and then speeds up as acid is generated.
Mechanism: Involves enol or enolate intermediates.
Hell-Volhard-Zelinsky reaction: Used for alpha bromination of carboxylic acids.
Alkylation of Enolates
Direct Alkylation
Enolates can react with alkyl halides via SN2 mechanisms to form new C–C bonds. Primary alkyl halides are preferred to avoid elimination side reactions.
Limitations: Secondary and tertiary halides often lead to elimination rather than substitution.
Acetoacetic ester synthesis: A milder method for alkylating enolates, using esters such as ethyl acetoacetate.
Acetoacetic Ester and Malonic Ester Synthesis
Formation of Ketones and Carboxylic Acids
These classic synthetic methods use the acidity of methylene groups between two carbonyls to generate enolates, which can then be alkylated and hydrolyzed to yield ketones or carboxylic acids.
Acetoacetic ester synthesis: Produces substituted ketones.
Malonic ester synthesis: Produces substituted carboxylic acids.
Enamine Chemistry
Enamines as Nucleophiles
Enamines, formed from secondary amines and carbonyl compounds, are less basic and more nucleophilic than enolates. They are useful for alkylation and acylation reactions, and can be hydrolyzed back to carbonyl compounds after reaction.
Stork enamine synthesis: Developed by Gilbert Stork, this method allows for selective alkylation of ketones.
Claisen Condensation
Formation of 1,3-Dicarbonyl Compounds
The Claisen condensation involves the reaction of two esters (or one ester and one ketone) in the presence of a base to form a β-keto ester or β-diketone. Deprotonation of the product is essential for the reaction to proceed.
Dieckmann condensation: An intramolecular version that forms five- or six-membered rings.
Crossed Claisen condensation: Involves two different esters, one of which must lack alpha hydrogens to avoid self-condensation.
Aldol Reaction
Base- and Acid-Catalyzed Aldol Reactions
The aldol reaction forms β-hydroxy carbonyl compounds from enolates and aldehydes or ketones. Under basic conditions, the reaction is reversible and can be isolated for aldehydes. Acid-catalyzed reactions typically proceed to dehydration, forming α,β-unsaturated carbonyl compounds.
Crossed aldol reactions: Use two different carbonyl compounds; selectivity is achieved by controlling enolate formation.
Dehydration: Drives the reaction forward, especially for ketone aldols.
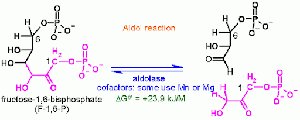
Conjugate (Michael) Addition
1,2- vs. 1,4-Addition
Enolates and other nucleophiles can add to α,β-unsaturated carbonyl compounds. Hard nucleophiles (organolithium, Grignard reagents) favor 1,2-addition, while soft nucleophiles (organocuprates) favor 1,4-addition (Michael addition).
Michael addition: Key method for forming C–C bonds at the β-position of enones and related compounds.
Cuprate reagents: Allow for selective 1,4-addition and further functionalization.
Robinson Annulation
Formation of Six-Membered Rings
The Robinson annulation combines a Michael addition and an intramolecular aldol condensation to construct six-membered rings, a key step in the synthesis of many natural products, including steroids.
Mechanism: Michael addition followed by aldol condensation and dehydration.
Biochemical Context: Fatty Acid Synthesis
Comparison of Biochemical and Organic Mechanisms
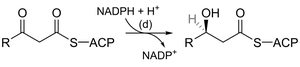
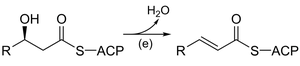
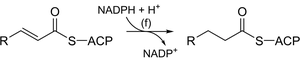
Fatty acid synthesis in biochemistry involves a series of reactions analogous to organic enolate chemistry, including Claisen condensation, reduction, dehydration, and further reduction. These steps are catalyzed by enzyme complexes and use cofactors such as NADPH.
Key enzymes: 3-ketoacyl-ACP synthase, 3-ketoacyl-ACP reductase, 3-hydroxyacyl-ACP dehydrase, enoyl-ACP reductase.
Comparison: The sequence of reactions mirrors laboratory organic synthesis but occurs under physiological conditions with enzyme catalysis.

Summary Table: Key Reactions of Alpha Carbon Chemistry
Reaction | Key Features | Typical Reagents/Conditions |
|---|---|---|
Enolate Formation | Deprotonation at alpha carbon | Strong base (LDA, NaOEt) |
Alpha Halogenation | Substitution of alpha hydrogen with halogen | Br2, Cl2, acid or base |
Alkylation | SN2 reaction with alkyl halide | Enolate + R–X |
Claisen Condensation | Formation of β-keto ester | 2 esters, base |
Aldol Reaction | Formation of β-hydroxy carbonyl | Enolate + aldehyde/ketone, base or acid |
Michael Addition | 1,4-addition to α,β-unsaturated carbonyl | Enolate or cuprate, enone |
Robinson Annulation | Michael addition + aldol condensation | Enolate, enone, base |
Practice Problems and Applications
Example Synthesis Problems
Design a synthesis of 3-ethyl-2-pentanone from ethyl acetoacetate.
Predict the product of a crossed aldol reaction between benzaldehyde and acetone.
Explain the selectivity in the Michael addition of a stabilized enolate to methyl vinyl ketone.
Additional info: The notes above integrate both the provided content and standard academic context for a comprehensive review of alpha carbon chemistry, enolate reactivity, and their applications in organic synthesis and biochemistry.
