 Back
BackAmides, Step-Growth Polymers, and Spectroscopy of Carboxylic Acid Derivatives
Study Guide - Smart Notes

Preparation and Reactions of Amides
Preparation of Amides from Carboxylic Acid Derivatives
Amides are important organic compounds that can be synthesized from carboxylic acid derivatives. Direct conversion from carboxylic acids is challenging due to the poor leaving ability of the hydroxyl group. Therefore, the carboxylic acid is first converted to a more reactive derivative, such as an acid chloride, before reaction with ammonia or an amine.
Activation of Carboxylic Acid: The hydroxyl group is replaced with a better leaving group, often using reagents like thionyl chloride (SOCl2) or dicyclohexylcarbodiimide (DCC).
Reaction with Ammonia or Amines: Acid chlorides react with ammonia (NH3), primary amines (1°), or secondary amines (2°) to form 1°, 2°, or 3° amides, respectively. Excess amine is used to neutralize the HCl byproduct.
General Equation:
Reactions of Amides
Hydrolysis: Amides can be hydrolyzed to carboxylic acids under acidic (H3O+, Δ) or basic (NaOH) conditions. The reaction is more difficult than for other carboxylic acid derivatives due to the stability of the amide bond.
Reduction: Amides are reduced by lithium aluminum hydride (LiAlH4) to amines, not alcohols. The reaction is followed by aqueous workup.
Example:
Step-Growth Polymers
Types and Synthesis
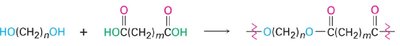
Step-growth polymers are formed by the stepwise reaction of bifunctional or multifunctional monomers. Polyamides and polyesters are two major classes, synthesized via nucleophilic acyl substitution reactions.
Polyamides (e.g., Nylon): Formed from diamines and diacid chlorides.
Polyesters (e.g., Dacron): Formed from diols and diacids.

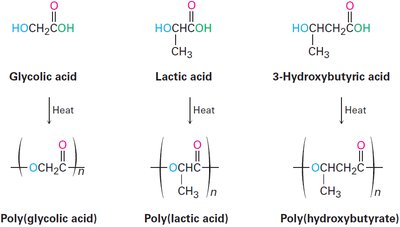
Biodegradable Polymers
Biodegradable polymers, such as polyglycolic acid, polylactic acid, and polyhydroxybutyrate, are susceptible to hydrolysis and break down rapidly in the environment. These are often used in medical and packaging applications.
Examples of Step-Growth Polymers
Nylon 66: Used in engineering and fibers.
Dacron (Mylar): Used in textiles and recording tape; has high tensile strength.
Spectroscopy of Carboxylic Acid Derivatives
Infrared (IR) Spectroscopy
IR spectroscopy is a key tool for identifying functional groups in organic molecules, especially carbonyl-containing compounds.
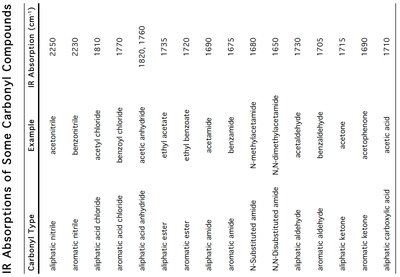
Carbonyl Stretch (C=O): Strong absorption between 1650–1850 cm-1.
Amide N–H Stretch: 3300–3500 cm-1. 1° amides show two bands, 2° amides one band, and 3° amides none in this region.
Characteristic Absorptions:
Acid chloride: ~1810 cm-1
Anhydride: ~1820, 1760 cm-1
Ester: ~1735 cm-1
Amide: 1690–1650 cm-1
Carboxylic acid: ~1710 cm-1
Example IR Spectra
Propanamide: Shows strong C=O and N–H stretches.
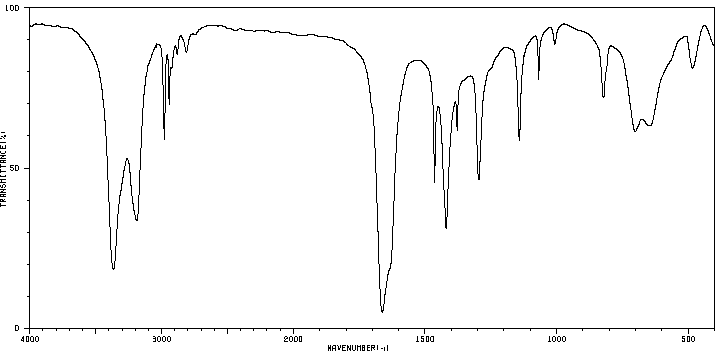
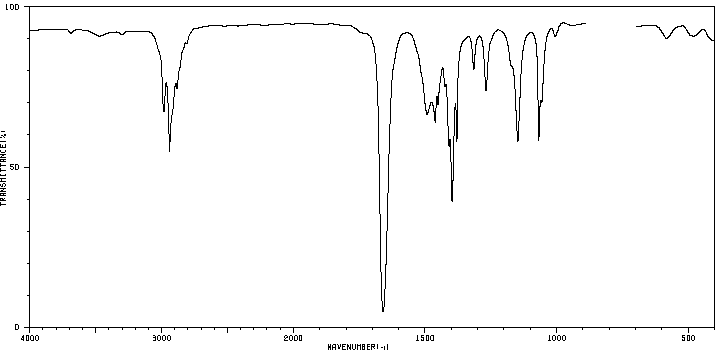
N-Methylpropanamide: Shows C=O and a single N–H stretch.
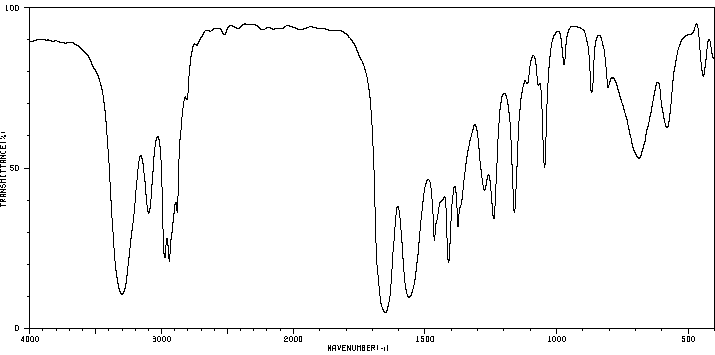
N,N-Dimethylpropanamide: Shows C=O stretch, but no N–H stretch.
1H NMR Spectroscopy
Nuclear Magnetic Resonance (NMR) spectroscopy provides information about the hydrogen environments in organic molecules.
Protons Adjacent to Carbonyl: Slightly deshielded, appear at δ 2.0–2.5 ppm.
Esters: Protons adjacent to oxygen are further downfield (δ 3.5–4.0 ppm).
Amides: Additional absorptions at δ 2.2–3.0 ppm (N–CH3) and δ 6.0–9.0 ppm (N–H, broad and variable).
Example 1H NMR Spectra
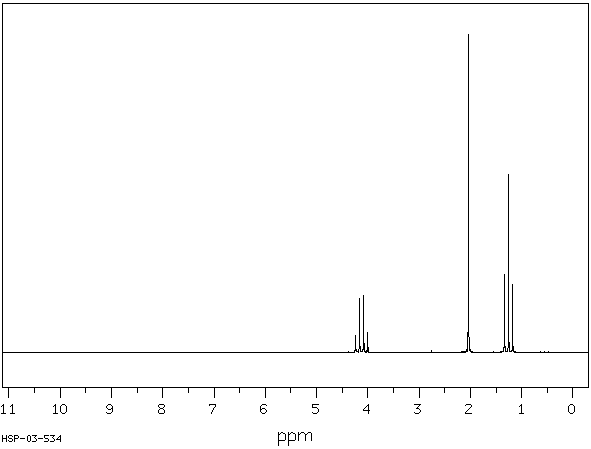
Ethyl Acetate: Shows characteristic ester signals.
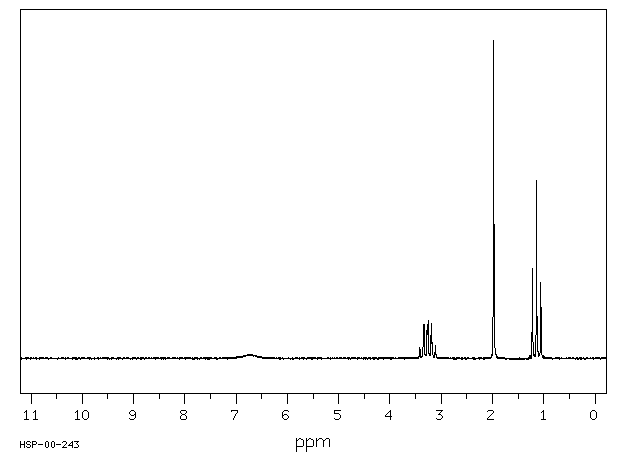
N-Ethylacetamide: Shows amide and ethyl group signals.
13C NMR Spectroscopy
Carbonyl carbons in carboxylic acid derivatives appear in the range δ 160–180 ppm. The exact position depends on the nature of the derivative (e.g., acid chloride, ester, amide, etc.).
Example Chemical Shifts:
Acid chloride: ~170 ppm
Ester: ~170–173 ppm
Amide: ~166–171 ppm
Carboxylic acid: ~177 ppm
Aldehyde/ketone: ~200 ppm
