 Back
BackAromaticity and Benzene: Structure, Stability, and Nomenclature
Study Guide - Smart Notes

Chapter 15: Benzene and Aromatic Compounds
Structure and Bonding in Benzene
Benzene is a prototypical aromatic compound, characterized by a planar, cyclic structure with delocalized π electrons. The unique stability of benzene arises from its electronic configuration and molecular orbital interactions.
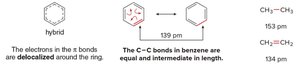
Delocalization of π Electrons: The electrons in the π bonds are delocalized around the ring, forming a continuous cloud of electron density above and below the plane of the ring.
Bond Lengths: The C–C bonds in benzene are equal and intermediate in length (139 pm), compared to single (153 pm) and double (134 pm) bonds in aliphatic compounds.
Hybrid Structure: Benzene is often represented as a resonance hybrid, indicating the equal distribution of electrons.
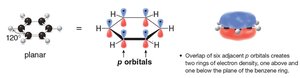
Planarity: Benzene is planar, with bond angles of 120°, allowing for optimal overlap of p orbitals.
p Orbital Overlap: Overlap of six adjacent p orbitals creates two rings of electron density, one above and one below the plane of the benzene ring.
Nomenclature of Benzene Derivatives
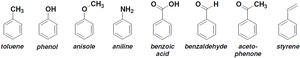
Benzene derivatives are named by identifying substituents and their positions on the ring. Many monosubstituted benzenes have common names, which are often used as parent names for more complex derivatives.
Monosubstituted Benzenes: Common names include toluene, phenol, aniline, anisole, benzoic acid, benzaldehyde, acetophenone, and styrene.
Disubstituted Benzenes: The prefixes ortho (1,2-), meta (1,3-), and para (1,4-) are used to designate relative positions of substituents.
Multiple Substituents: For three or more substituents, number the ring to give the lowest possible numbers, alphabetize substituent names, and use common roots when applicable.
Benzene as a Substituent: A benzene ring as a substituent is called a phenyl group (Ph−), while a benzyl group contains a benzene ring attached to a CH2 group.
Stability of Benzene and Aromaticity
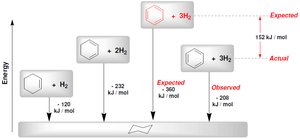
Benzene exhibits exceptional stability due to its aromatic nature, which can be quantified by heats of hydrogenation. Not all fully conjugated cyclic compounds are aromatic; some are reactive and lack aromatic stability.
Heats of Hydrogenation: Benzene's observed heat of hydrogenation is much less than expected, indicating extra stability.
Aromaticity Criteria: Aromatic compounds must be cyclic, planar, fully conjugated, and satisfy Hückel's rule (4n+2 π electrons).
Antiaromatic and Nonaromatic Compounds: Antiaromatic compounds are cyclic, planar, fully conjugated, but have 4n π electrons. Nonaromatic compounds lack one or more requirements for aromaticity.

Hückel's Rule and Classification of Aromatic Compounds
Hückel's rule provides a quantitative criterion for aromaticity based on the number of π electrons in the ring. Molecular orbital theory explains why only certain electron counts confer aromatic stability.
Hückel's Rule: A compound is aromatic if it has 4n+2 π electrons (n = 0, 1, 2, ...).
Classification:
Aromatic: Cyclic, planar, fully conjugated, 4n+2 π electrons.
Antiaromatic: Cyclic, planar, fully conjugated, 4n π electrons.
Nonaromatic: Lacks cyclic, planar, or fully conjugated structure.
Annulenes: Hydrocarbons with a single ring and alternating double/single bonds are called annulenes, named by the number of atoms in the ring (e.g., [10]-annulene).
Polycyclic Aromatic Hydrocarbons (PAHs): Fused rings form larger aromatic systems such as anthracene and phenanthrene.
Heterocycles: Rings containing O, N, or S can also be aromatic if they meet the criteria.
![[10]-annulene structure and non-aromaticity](https://static.studychannel.pearsonprd.tech/study_guide_files/organic-chemistry/sub_images/bc3a1677_image_14.png)
Cyclopentadienyl Compounds and Aromaticity
Cyclopentadienyl anion is aromatic due to its fully conjugated ring and 6 π electrons, while cyclopentadiene itself is not aromatic.
Cyclopentadiene: Not aromatic; pKa = 15.
Cyclopentadienyl Anion: Aromatic; stabilized conjugate base due to 6 π electrons.
Molecular Orbital Theory and Aromaticity
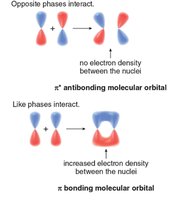
Molecular orbital (MO) theory explains the stability of aromatic compounds by the arrangement of bonding and antibonding orbitals. The combination of p orbitals can be constructive (bonding) or destructive (antibonding).
Bonding vs. Antibonding Orbitals: Like phases interact to form bonding orbitals; opposite phases form antibonding orbitals.
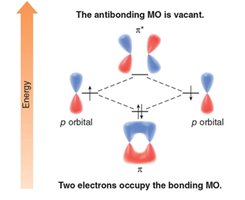
HOMO and LUMO: The highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) are key concepts in MO theory.
Degenerate Orbitals: Orbitals with the same energy are called degenerate.
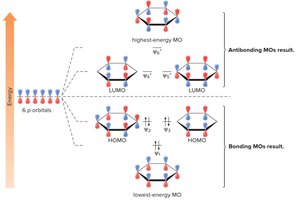
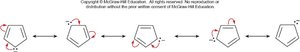
Inscribed Polygon Method for Predicting Aromaticity
The inscribed polygon method is a graphical approach to predict aromaticity by visualizing molecular orbitals and electron filling.
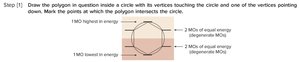
Step 1: Draw the polygon inside a circle, with vertices touching the circle and one vertex pointing down. Mark the points where the polygon intersects the circle.
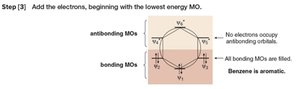
Step 3: Add electrons starting from the lowest energy MO. Benzene is aromatic because all bonding MOs are filled and no electrons occupy antibonding orbitals.
Summary Table: Classification of Cyclic Compounds by Aromaticity
Type | Criteria | Example | π Electrons |
|---|---|---|---|
Aromatic | Cyclic, planar, fully conjugated, 4n+2 π electrons | Benzene | 6 |
Antiaromatic | Cyclic, planar, fully conjugated, 4n π electrons | Cyclobutadiene | 4 |
Nonaromatic | Not cyclic, not planar, or not fully conjugated | Cyclooctatetraene | 8 |
Example: Benzene is aromatic because it is cyclic, planar, fully conjugated, and has 6 π electrons (n=1 in Hückel's rule).
Additional info: The notes include expanded explanations of molecular orbital theory and the inscribed polygon method for predicting aromaticity, which are essential for understanding the electronic structure and stability of aromatic compounds.
