 Back
BackAromaticity: Structure, Stability, and Reactivity of Aromatic Compounds
Study Guide - Smart Notes

Aromaticity
Introduction to Aromaticity
Aromaticity is a fundamental concept in organic chemistry, describing the unusual stability and unique reactivity of certain cyclic, planar molecules with conjugated pi electron systems. The archetype of aromatic compounds is benzene, which exhibits properties distinct from typical alkenes due to its delocalized electrons.
Aromatic compounds are more stable than expected from simple resonance or conjugation.
They do not react like typical alkenes and resist addition reactions that would disrupt their aromatic system.
The term "aromatic" originally referred to fragrant compounds but now refers to this special electronic structure.

Defining Aromatic Compounds
To be classified as aromatic, a compound must satisfy several criteria:
Planarity: The molecule must be flat, allowing for continuous overlap of p orbitals.
Conjugation: There must be a cyclic system of contiguous (adjacent) p orbitals.
Hückel’s Rule: The molecule must have 4n + 2 pi electrons (where n is a non-negative integer).
Complete filling of bonding molecular orbitals: All bonding pi orbitals must be filled.
Unusual stability: Aromatic compounds are more stable than similar non-aromatic or anti-aromatic compounds.
Ring current: Aromatic compounds support a ring current, detectable by NMR spectroscopy.

Models of Aromatic Bonding
Several models are used to describe the bonding in aromatic compounds, each with its own strengths and limitations:
Localized Bonding Model: Predicts alternating single and double bonds, but fails to explain the observed equivalence of all bonds in benzene and the absence of isomers for 1,2-disubstituted benzenes.
Resonance Model: Uses resonance structures to explain bond equivalence and stability, but underestimates the stabilization energy of benzene.
Molecular Orbital (MO) Model: Describes the delocalization of pi electrons over the entire ring, accounting for the extra stability and unique reactivity of aromatic compounds.
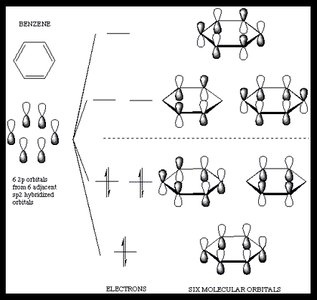
Molecular Orbitals in Aromatic Systems
The MO model explains aromaticity by the formation of bonding and antibonding molecular orbitals from the combination of atomic p orbitals:
In benzene, six p orbitals combine to form six molecular orbitals: three bonding and three antibonding.
The six pi electrons fill the three lowest-energy bonding orbitals, resulting in a closed-shell, highly stable system.
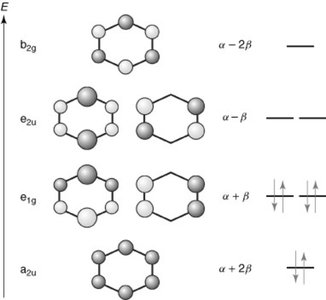

Hückel’s Rule and Annulenes
Hückel’s Rule states that a planar, monocyclic, fully conjugated polyene is aromatic if it contains 4n + 2 pi electrons. This rule helps classify annulenes (cyclic polyenes) as aromatic, anti-aromatic, or non-aromatic:
Annulene | Electron Count | Aromaticity |
|---|---|---|
[4]annulene | 4 | Anti-aromatic |
[6]annulene (benzene) | 6 | Aromatic |
[8]annulene | 8 | Anti-aromatic |
[10]annulene | 10 | Aromatic (if planar) |
[12]annulene | 12 | Anti-aromatic |
[14]annulene | 14 | Aromatic |
[16]annulene | 16 | Anti-aromatic |
[18]annulene | 18 | Aromatic |
Additional info: Actual aromaticity may be affected by non-planarity or steric hindrance in larger annulenes.
Aromatic Ions and Heteroaromatics
Aromaticity is not limited to neutral hydrocarbons. Ions and heterocycles can also be aromatic if they meet the criteria:
Cyclopentadienyl anion (C5H5-): Aromatic with 6 pi electrons.
Tropylium cation (C7H7+): Aromatic with 6 pi electrons.
Heteroaromatics: Compounds like pyridine, pyrrole, and imidazole contain heteroatoms (N, O, S) that contribute lone pairs to the aromatic system.

Heterocyclic Aromatic Compounds
Heterocycles are rings containing atoms other than carbon. Their aromaticity depends on the hybridization and electron contribution of the heteroatom:
Pyridine: Nitrogen is sp2 hybridized; its lone pair is not part of the aromatic pi system.
Pyrrole: Nitrogen is sp2 hybridized; its lone pair is part of the aromatic pi system, making it less basic.
Imidazole: Contains two nitrogens, one with a lone pair in the aromatic system and one with a basic lone pair orthogonal to the ring.



Polycyclic Aromatic Compounds (PACs)
Fused aromatic rings, such as naphthalene, anthracene, and phenanthrene, are also aromatic. Their stability and reactivity can differ from benzene due to the distribution of pi electrons and the presence of more reactive bonds.
Naphthalene: 10 pi electrons, aromatic but not twice as stabilized as benzene.
Phenanthrene: 14 pi electrons, overall aromatic but with one more reactive bond.
Benzo[a]pyrene: A polycyclic aromatic hydrocarbon (PAH) found in soot and smoke, known for its carcinogenic properties.

Reactivity of Aromatic Compounds
Aromatic compounds typically undergo substitution reactions rather than addition, preserving the aromatic system. Some important reactions include:
Benzylic Halogenation: Halogenation at the benzylic position proceeds via SN1 or SN2 mechanisms due to resonance stabilization of the benzylic carbocation.
Benzylic Oxidation: Oxidation of benzylic positions (with a benzylic hydrogen) using strong oxidants like KMnO4 yields carboxylic acids.
Birch Reduction: Partial reduction of aromatic rings using sodium in liquid ammonia produces non-aromatic cyclohexadienes.

Birch Reduction Mechanism
The Birch reduction involves the addition of electrons and protons to an aromatic ring, resulting in a 1,4-cyclohexadiene. The mechanism proceeds via radical anion intermediates and is selective for certain substituents.
Works for alkyl and alkoxy-substituted benzenes, but not for esters, ketones, or aldehydes (which are reduced instead).
Protonation occurs at positions of highest electron density.
Naming Aromatic Compounds
Common and systematic nomenclature is used for aromatic compounds:
Ortho- (o-), meta- (m-), para- (p-): Prefixes for 1,2-, 1,3-, and 1,4-disubstituted benzenes, respectively.
For more than two substituents, numbers are used to indicate positions, and substituents are listed alphabetically.
Summary Table: Aromaticity Criteria
Criterion | Description |
|---|---|
Planarity | All atoms in the ring are in the same plane |
Conjugation | Continuous overlap of p orbitals |
Electron Count | 4n + 2 pi electrons (Hückel’s Rule) |
Bonding Orbitals | All bonding pi orbitals filled |
Stability | Unusual thermodynamic stability |
Ring Current | Detectable by NMR |
Anion/Heteroatom Contribution | Anions and heteroatoms can contribute electrons |
Additional info: Aromaticity is a key concept in understanding the structure, reactivity, and properties of many organic molecules, including biologically important compounds and industrial chemicals.
