 Back
BackCarboxylic Acids and Nitriles: Structure, Properties, Acidity, and Synthesis
Study Guide - Smart Notes

Carboxylic Acids and Nitriles
Overview of Functional Groups
Carboxylic acids and nitriles are important organic functional groups characterized by a carbon atom bonded to electronegative atoms and containing a π bond. Both groups are electrophilic and undergo nucleophilic addition reactions. Carboxylic acids are precursors to many other functional groups, known as carboxylic acid derivatives, and are widespread in nature, participating in numerous biological pathways.
Carboxylic Acid: Contains three bonds to oxygen (RCOOH).
Nitrile: Contains three bonds to nitrogen (RCN).
Carboxylic Acid Derivatives: Include acid chlorides, esters, amides, and thioesters.
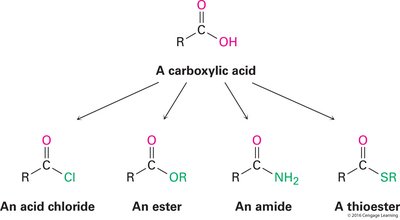
Additional info: Carboxylic acids are present in almost all biological pathways and serve as key intermediates in organic synthesis.
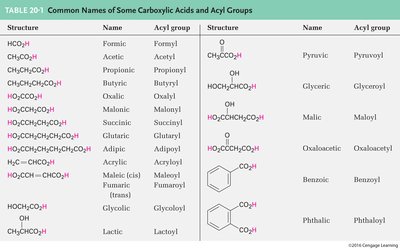
Common Carboxylic Acids and Their Acyl Groups
Carboxylic acids are named based on their parent alkane, with the terminal '-e' replaced by '-oic acid.' The carbon of the carboxylic acid is always C1. When attached to a ring, the group is named as a 'carboxylic acid,' and the substituent is known as a carboxyl group.
Structure | Name | Acyl group |
|---|---|---|
HCO2H | Formic | Formyl |
CH3CO2H | Acetic | Acetyl |
CH3CH2CO2H | Propionic | Propionyl |
CH3CH2CH2CO2H | Butyric | Butyryl |
HOOCCH2CO2H | Malonic | Malonyl |
HOOCCH2CH2CO2H | Succinic | Succinyl |
HOOCCH2CH2CH2CO2H | Glutaric | Glutaryl |
HOOCCH=CHCO2H | Acrylic | Acryloyl |
HOCH2CO2H | Glycolic | Glycolyl |
CH3CHOHCO2H | Lactic | Lactoyl |
PhCO2H | Benzoic | Benzoyl |
Structure and Properties of Carboxylic Acids
Carboxylic acids have a basic structure that allows them to form hydrogen bonds, which significantly influences their boiling points. Hydrogen bonding also affects their solubility and physical properties.
Hydrogen Bonding: Leads to higher boiling points compared to similar molecular weight compounds.
Solubility: Carboxylic acids with fewer than six carbons are generally water-soluble; larger acids are less soluble.
Acidity of Carboxylic Acids
Carboxylic acids are acidic and react with bases to form carboxylate salts, which are more water-soluble. The acidity is quantified by the acidity constant () and the value. Most carboxylic acids have a $pK_a$ around 4.5, making them much more acidic than alcohols.
Acidity Constant (): Measures the extent of dissociation in aqueous solution.
Carboxylate Ion Stability: Resonance stabilization makes carboxylate ions more stable than alkoxide ions.
Comparison: Carboxylic acids are more acidic than alcohols due to resonance stabilization of the carboxylate ion.
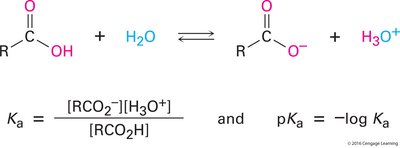
Additional info: The is calculated as .
Substituent Effects on Acidity
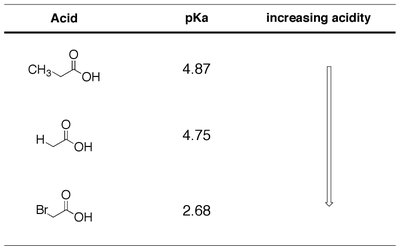
The acidity of carboxylic acids is influenced by substituents attached to the molecule. Electron-withdrawing groups increase acidity by stabilizing the carboxylate ion, while electron-donating groups decrease acidity.
Aliphatic Acids: Substituents such as halogens increase acidity.
Additivity: Multiple electron-withdrawing groups further increase acidity.
Proximity: The closer the substituent to the carboxyl group, the greater its effect.
Benzoic Acids: Substituent effects are also observed in aromatic carboxylic acids.
Acid | pKa | Increasing Acidity |
|---|---|---|
CH3COOH | 4.87 | ↓ |
HCOOH | 4.75 | |
BrCH2COOH | 2.68 |
Acid | pKa | Increasing Acidity |
|---|---|---|
CH3COOH | 4.75 | ↓ |
ClCH2COOH | 2.85 | |
Cl2CHCOOH | 1.48 | |
Cl3CCOOH | 0.64 |
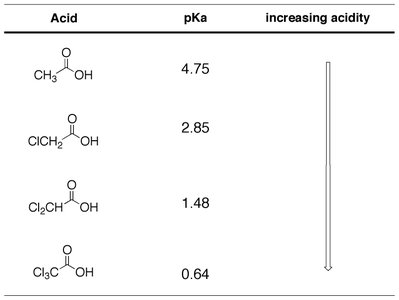
Acid | pKa | Increasing Acidity |
|---|---|---|
CH3COOH | 4.82 | ↓ |
ClCH2CH2COOH | 4.52 | |
ClCH2COOH | 4.05 | |
ClCH2COOH (closer Cl) | 2.86 |
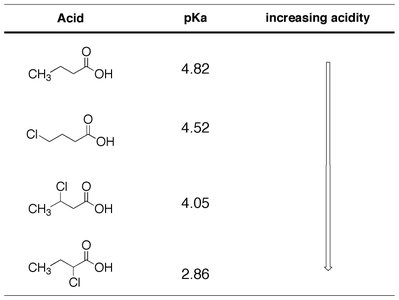
Group (G) | pKa | Increasing Acidity |
|---|---|---|
OH | 4.48 | ↓ |
OCH3 | 4.46 | |
CH3 | 4.34 | |
H (benzoic acid) | 4.19 | |
Cl | 4.00 | |
Br | 3.96 | |
CHO | 3.75 | |
CN | 3.55 | |
NO2 | 3.41 |
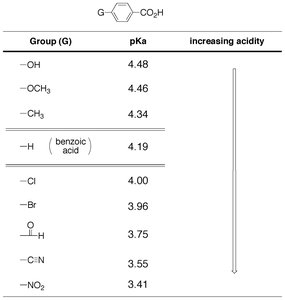
Additional info: Electron-withdrawing groups stabilize the carboxylate ion, increasing acidity, while electron-donating groups destabilize it, decreasing acidity.
Preparation of Carboxylic Acids
Carboxylic acids can be synthesized by several methods, each with specific requirements and outcomes:
Oxidation of Alkyl Benzenes: Requires a benzylic hydrogen; uses KMnO4.
Oxidative Cleavage of Alkenes: KMnO4 oxidizes alkenes to carboxylic acids.
Oxidation of 1° Alcohols and Aldehydes: Jones reagent (CrO3/H2SO4) or AgNO3/NH4OH.
Hydrolysis of Nitriles: Acidic or basic hydrolysis extends the carbon chain by one.
Carboxylation of Grignard Reagents: Reaction with CO2 extends the carbon chain by one.
Example: Synthesis of carboxylic acids from alkyl halides via Grignard reagents or nitrile hydrolysis.
Additional info: Synthetic design often involves choosing between nitrile hydrolysis and Grignard carboxylation for chain extension.
Reactions of Carboxylic Acids
Carboxylic acids undergo several important reactions:
Deprotonation: Readily deprotonated by bases (NaOH, NaHCO3, NaH, RMgX, RLi, RNH2) to form carboxylate salts.
Reduction: Reduced by LiAlH4 (non-selective) or BH3 (selective for carboxylic acids over esters) to primary alcohols.
Example: Selective reduction of carboxylic acids in the presence of esters using BH3.
Additional info: LiAlH4 reduces carboxylic acids, aldehydes, ketones, and esters, while BH3 is more selective for carboxylic acids.
