 Back
BackChapter 17: Introduction to Carbonyl Chemistry and Organometallic Reactions
Study Guide - Smart Notes

Introduction to Carbonyl Chemistry
Classification of Carbonyl Compounds
Carbonyl compounds are organic molecules containing a carbon-oxygen double bond (C=O). They are classified based on the atoms or groups attached to the carbonyl carbon:
Compounds with only carbon and hydrogen atoms bonded to the carbonyl (e.g., aldehydes and ketones).
Compounds with an electronegative atom (such as O, N, or Cl) bonded to the carbonyl (e.g., carboxylic acids, esters, amides, acid chlorides).
General Reactions of Carbonyl Compounds
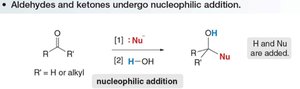
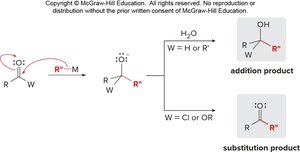
Nucleophilic Addition to Aldehydes and Ketones
Aldehydes and ketones undergo nucleophilic addition reactions, where a nucleophile attacks the electrophilic carbonyl carbon, followed by protonation to yield an alcohol.
Step 1: Nucleophile adds to the carbonyl carbon.
Step 2: Protonation of the oxygen atom forms an alcohol.
Reactivity: Aldehydes are generally more reactive than ketones due to less steric hindrance and less electron-donating alkyl groups.

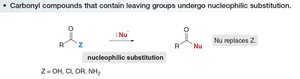
Nucleophilic Substitution in Carbonyl Compounds with Leaving Groups
Carbonyl compounds containing a good leaving group (Z) undergo nucleophilic acyl substitution, where the nucleophile replaces the leaving group.
Common leaving groups: OH, Cl, OR, NH2
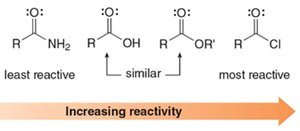
Reactivity order: Acid chlorides > esters ≈ carboxylic acids > amides
Oxidation and Reduction of Carbonyl Compounds
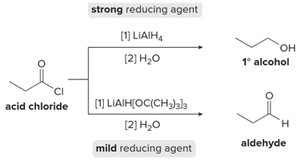
Reduction of Carbonyl Compounds
Reduction reactions convert carbonyl groups to alcohols or amines. The choice of reducing agent determines the product:
Strong reducing agents (e.g., LiAlH4) reduce acid chlorides to primary alcohols.
Mild reducing agents (e.g., LiAlH[OC(CH3)3]3) can selectively reduce acid chlorides to aldehydes.

Oxidation of Aldehydes
Aldehydes can be oxidized to carboxylic acids using various oxidizing agents:
Common oxidants: CrO3, Na2Cr2O7, K2Cr2O7, KMnO4
Selective oxidation: Tollen’s reagent (Ag2O in NH4OH) oxidizes aldehydes in the presence of other functional groups.
Organometallic Reagents in Carbonyl Chemistry
Preparation of Organocuprate Compounds and Acetylide Ions
Organometallic reagents are powerful nucleophiles used to form new carbon-carbon bonds:
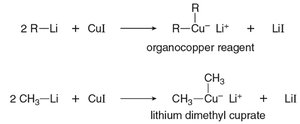
Organocuprates (Gilman reagents): Prepared by reacting organolithium compounds with CuI.
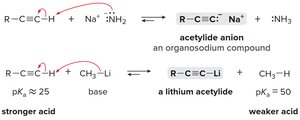
Acetylide ions: Generated by deprotonating terminal alkynes with strong bases (e.g., NaNH2 or organolithium reagents).
Reactivity and Transformations of Organometallic Compounds
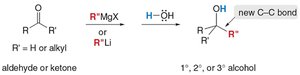
Organometallic reagents react with carbonyl compounds to form alcohols or other functional groups, depending on the substrate and reagent.
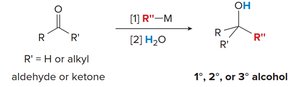
Grignard and organolithium reagents: Add to aldehydes and ketones to form 1°, 2°, or 3° alcohols.
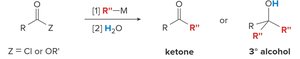
With esters or acid chlorides: Two equivalents are required to form tertiary alcohols.
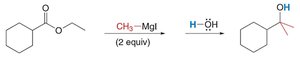
Limitations of Organometallic Reagents
Organometallic reagents are highly reactive and cannot be used with molecules containing both a carbonyl group and N—H or O—H bonds, as they will react with the acidic hydrogen instead of the carbonyl.
Reactions with α,β-Unsaturated Carbonyl Compounds
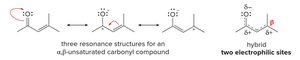
Structure and Reactivity
α,β-Unsaturated carbonyl compounds have conjugated double bonds, resulting in two electrophilic sites (the carbonyl carbon and the β-carbon). Resonance structures illustrate the delocalization of electrons.
1,2- vs. 1,4-Addition to α,β-Unsaturated Carbonyls
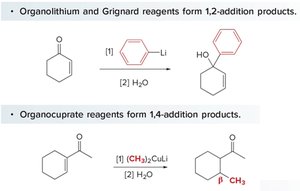
Different organometallic reagents add to different positions:
Organolithium and Grignard reagents: Prefer 1,2-addition (to the carbonyl carbon).
Organocuprate reagents: Prefer 1,4-addition (to the β-carbon).
Summary of Organometallic Reactions
The outcome of reactions between organometallic reagents and carbonyl compounds depends on the nature of both the reagent and the substrate. Addition or substitution products can be obtained based on the presence of leaving groups and the type of organometallic reagent used.
Key Equations
Nucleophilic Addition:
Nucleophilic Acyl Substitution:
Reduction of Acid Chloride:
Oxidation of Aldehyde:
Organocuprate Formation:
Grignard Addition to Carbonyl:
Additional info: The above notes integrate foundational concepts of carbonyl chemistry, nucleophilic addition and substitution, oxidation/reduction, and the use of organometallic reagents, as covered in a typical college-level organic chemistry course.
