Back
BackCHEM 211: Organic Chemistry 1 – Exam 2 Review Study Notes
Study Guide - Smart Notes

Cycloalkanes and Their Nomenclature
Cycloalkane Nomenclature
Cycloalkanes are saturated hydrocarbons containing carbon atoms arranged in a ring. The nomenclature of cycloalkanes follows IUPAC rules, where the ring is considered the parent hydrocarbon, and substituents are named as alkyl groups attached to the ring.
Main Chain: The cycloalkane ring is the main chain; alkyl groups attached are named as substituents.
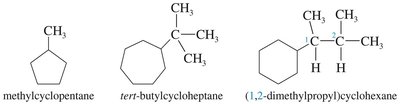
Numbering: If only one substituent is present, no number is necessary. For multiple substituents, numbering starts at the substituent that comes first alphabetically and proceeds to give the lowest possible numbers to the substituents.
Examples: Methylcyclopentane, tert-butylcycloheptane, (1,2-dimethylpropyl)cyclohexane.
Structures of Small Cycloalkanes
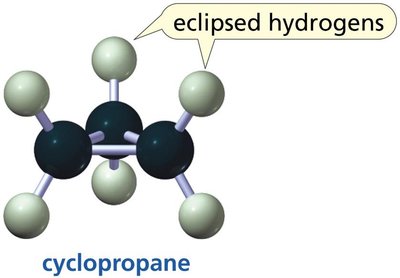
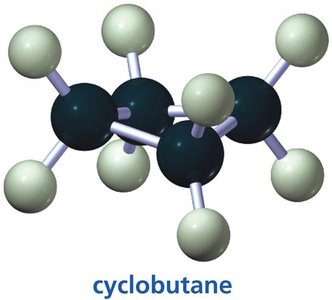
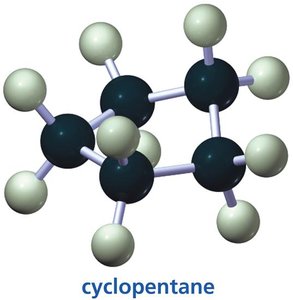
The three smallest cycloalkanes are cyclopropane, cyclobutane, and cyclopentane. Their structures are important for understanding ring strain and conformational analysis.
Cyclopropane: Planar, with significant angle strain (60° bond angles) and eclipsed hydrogens, leading to high reactivity.
Cyclobutane: Slightly puckered to reduce torsional strain, bond angles are about 88°.
Cyclopentane: Adopts an envelope conformation to minimize torsional strain, bond angles are close to 108°.
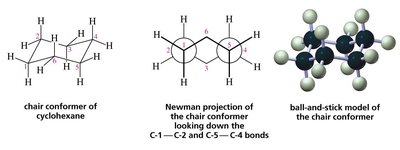
Chair Conformation of Cyclohexane
Cyclohexane adopts a chair conformation to minimize both angle and torsional strain, making it the most stable conformation. The chair form allows for staggered hydrogens and nearly ideal bond angles (109.5°).
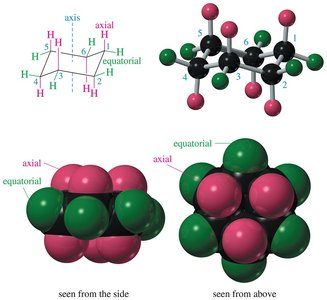
Chair Conformer: The most stable conformation of cyclohexane, with alternating axial and equatorial positions for hydrogens.
Newman Projection: Used to visualize the staggered arrangement of bonds in the chair conformer.
Axial and Equatorial Positions
In the chair conformation, each carbon has one axial and one equatorial hydrogen. Axial positions are perpendicular to the ring, while equatorial positions are roughly in the plane of the ring. Substituents prefer the equatorial position to minimize 1,3-diaxial interactions (steric hindrance).
Axial Bonds: Directed vertically, parallel to the axis of the ring.
Equatorial Bonds: Directed outward, toward the equator of the molecule.
The Study of Chemical Reactions
Thermodynamics, Kinetics, and Mechanism
Understanding organic reactions requires knowledge of thermodynamics (energy changes), kinetics (reaction rates), and mechanisms (stepwise pathways).
Thermodynamics: Study of energy changes during chemical and physical transformations.
Kinetics: Study of reaction rates and factors affecting them.
Mechanism: Step-by-step description of how a reaction occurs.
Homolytic and Heterolytic Cleavages
Bond cleavage can occur in two ways: homolytic (each atom gets one electron, forming radicals) and heterolytic (one atom gets both electrons, forming ions).
Homolytic Cleavage: Produces free radicals; common in radical reactions.
Heterolytic Cleavage: Produces ions; common in polar reactions.
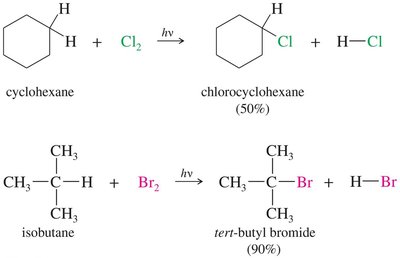
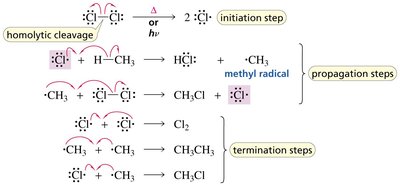
Chlorination of Methane: A Radical Chain Reaction
Chlorination of methane is a classic example of a radical chain reaction, requiring heat or light for initiation. The reaction proceeds via initiation, propagation, and termination steps.
Initiation: Formation of chlorine radicals by homolytic cleavage.
Propagation: Chlorine radicals react with methane to form methyl radicals and HCl, then methyl radicals react with Cl2 to regenerate chlorine radicals.
Termination: Combination of radicals to form stable products.


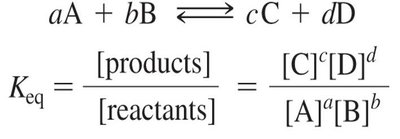
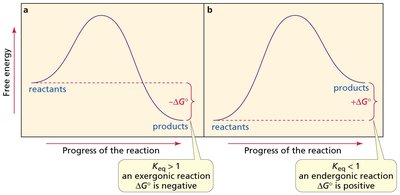
Equilibrium Constant and Free Energy Change
The equilibrium constant () expresses the ratio of product to reactant concentrations at equilibrium. The standard free energy change () relates to and determines reaction spontaneity.
Equilibrium Constant:
Free Energy Change:
Spontaneity: Negative indicates a spontaneous (exergonic) reaction; positive indicates a nonspontaneous (endergonic) reaction.
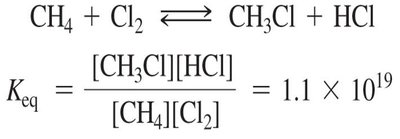
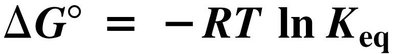
Kinetics and Reaction Coordinate Diagrams
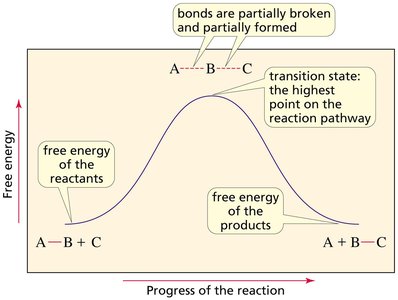
Kinetics studies the rate at which reactions proceed. Reaction coordinate diagrams illustrate the energy changes during a reaction, showing reactants, products, transition states, and intermediates.
Rate Law: Expresses the relationship between reactant concentrations and reaction rate; determined experimentally.
Transition State: The highest energy point along the reaction pathway.
Rate-Limiting Step: The slowest step with the highest activation energy ().
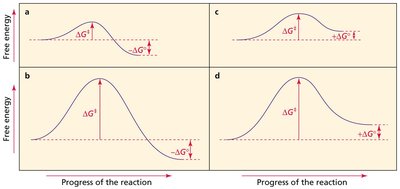
Hammond Postulate
The Hammond postulate states that the structure of a transition state resembles the structure of the nearest stable species. In endothermic reactions, the transition state is product-like; in exothermic reactions, it is reactant-like.
Endothermic Reaction: Transition state resembles the product.
Exothermic Reaction: Transition state resembles the reactant.
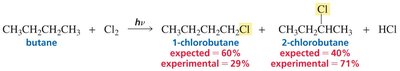
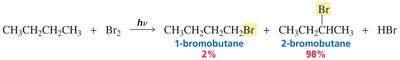
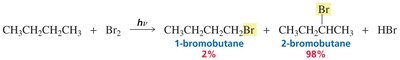
Chlorination vs Bromination: Reactivity–Selectivity Principle
Chlorine and bromine radicals differ in reactivity and selectivity. Bromine is less reactive but more selective, favoring substitution at more substituted carbons.
Chlorination: Less selective, more reactive; produces a mixture of products.
Bromination: More selective, less reactive; favors substitution at tertiary carbons.
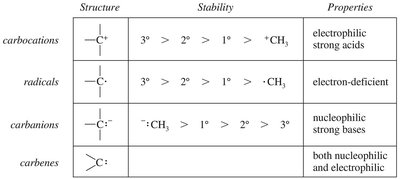
Summary of Reactive Species
Organic reactions often involve reactive intermediates such as carbocations, radicals, carbanions, and carbenes. Their stability and properties influence reaction pathways.
Structure | Stability | Properties |
|---|---|---|
Carbocations | 3° > 2° > 1° > methyl | Electrophilic, strong acids |
Radicals | 3° > 2° > 1° > methyl | Electron-deficient |
Carbanions | Methyl > 1° > 2° > 3° | Nucleophilic, strong bases |
Carbenes | - | Both nucleophilic and electrophilic |
Stereochemistry
Chiral Molecules and Asymmetric Centers
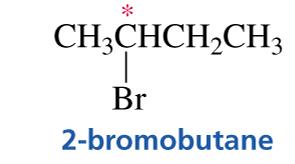
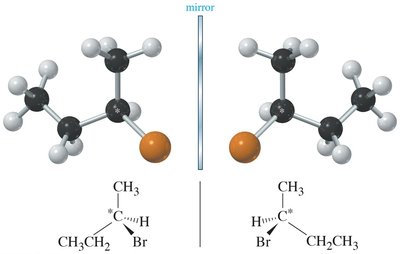
A molecule is chiral if it is not superimposable on its mirror image. Chirality arises from the presence of an asymmetric center—an atom attached to four different groups.
Chiral Molecule: Has an asymmetric center and no plane of symmetry.
Asymmetric Center: Typically a carbon atom bonded to four different groups.
Stereoisomers: Enantiomers and Diastereomers
Stereoisomers have the same connectivity but differ in the spatial arrangement of atoms. Enantiomers are non-superimposable mirror images; diastereomers are not mirror images.
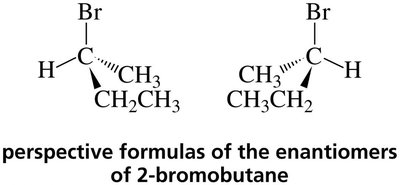
Enantiomers: Mirror images, opposite configuration at all chiral centers.
Diastereomers: Stereoisomers that are not mirror images; differ at some but not all chiral centers.
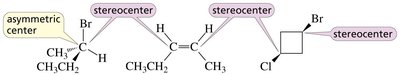
Asymmetric Center vs Stereocenter
All asymmetric centers are stereocenters, but not all stereocenters are asymmetric centers. A stereocenter is any atom at which the interchange of two groups produces a stereoisomer.
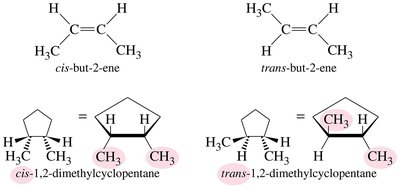
Geometric Isomers
Geometric (cis-trans) isomers arise from restricted rotation, typically around double bonds or in rings. They differ in the relative positions of substituents.
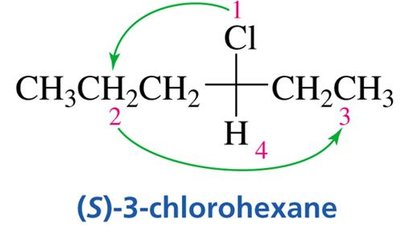
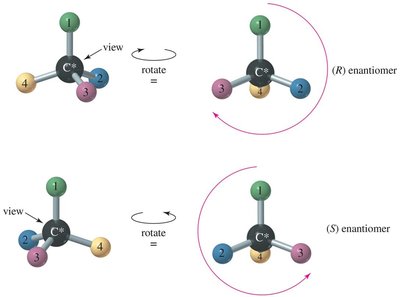
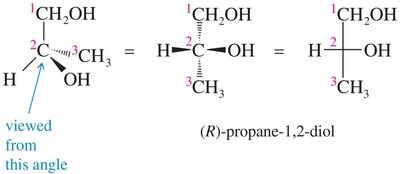
(R) and (S) Configuration: Cahn-Ingold-Prelog Convention
The Cahn-Ingold-Prelog (CIP) system assigns priorities to groups attached to a chiral center to distinguish between (R) and (S) configurations.
Step 1: Assign priorities based on atomic number; higher atomic number = higher priority.
Step 2: Orient the molecule so the lowest priority group is in the back; trace a path from highest to lowest priority.
Clockwise: (R) configuration; Counterclockwise: (S) configuration.
Multiple Bonds: Treat as if each bond is to a separate atom.
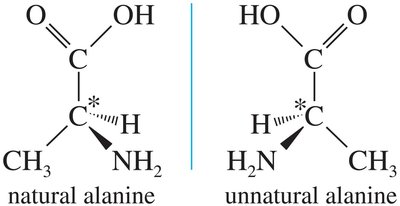
Properties of Enantiomers
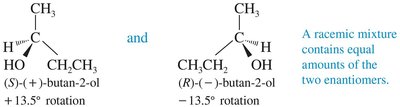
Enantiomers have identical physical properties (boiling point, melting point, density) except for their interaction with plane-polarized light and other chiral substances. They rotate plane-polarized light in equal but opposite directions.
Dextrorotatory (+): Rotates light clockwise.
Levorotatory (–): Rotates light counterclockwise.
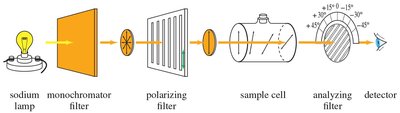
Optical Activity: Measured using a polarimeter.
Racemic Mixtures
A racemic mixture contains equal amounts of both enantiomers and is optically inactive due to the cancellation of optical rotations. Racemates may have different physical properties from pure enantiomers.
Drawing Enantiomers: Perspective and Fischer Projections
Enantiomers can be represented using perspective formulas or Fischer projections. Fischer projections are flat representations where vertical lines are bonds going away from the viewer and horizontal lines are bonds coming toward the viewer.

Compounds with Multiple Chiral Centers and Meso Compounds
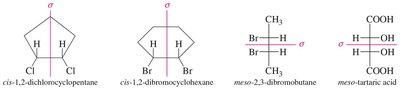
Compounds with two or more chiral centers can have enantiomers, diastereomers, or meso isomers. Meso compounds have a plane of symmetry and are achiral despite having chiral centers.
Maximum Number of Stereoisomers: , where n = number of chiral centers.
Meso Compounds: Achiral due to internal symmetry; superimposable on their mirror image.
Alkyl Halides and Nucleophilic Substitution
Classes of Alkyl Halides
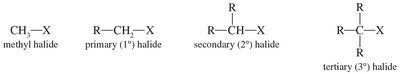
Alkyl halides are compounds in which a halogen is bonded to an sp3 carbon. They are classified based on the carbon to which the halogen is attached.
Alkyl Halides: Halogen bonded to sp3 carbon.
Vinyl Halides: Halogen bonded to sp2 carbon of an alkene.
Aryl Halides: Halogen bonded to sp2 carbon on a benzene ring.
Methyl, Primary, Secondary, Tertiary: Based on the number of carbons attached to the carbon bearing the halogen.

Preparation of Alkyl Halides
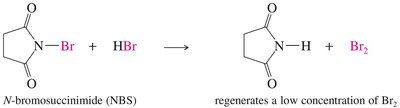
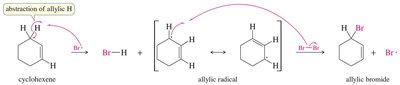
Alkyl halides can be prepared by free radical halogenation. Chlorination is less selective, while bromination is highly selective for more substituted carbons. Allylic halogenation introduces a halogen at the allylic position (adjacent to a double bond).
Chlorination: Produces a mixture of products; not ideal for selective synthesis.
Bromination: Highly selective; tertiary > secondary > primary carbons.
Allylic Halogenation: Halogen is introduced at the allylic position, often using N-bromosuccinimide (NBS).