 Back
BackChirality and Stereochemistry: An Introduction to Enantiomers and Stereoisomers
Study Guide - Smart Notes

Chirality and Enantiomers
Introduction to Chirality
Chirality is a fundamental concept in organic chemistry, describing objects or molecules that are not superimposable on their mirror images. This property is crucial for understanding the behavior of many organic compounds, especially in biological systems.
Chiral objects are those that cannot be superimposed on their mirror images, much like left and right hands.
Achiral objects can be superimposed on their mirror images, such as a fork or a cup.
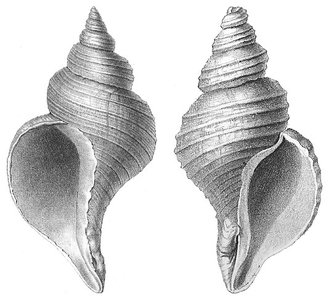
Chirality is observed in nature, for example, in the shells of certain sea snails, which exist in left-handed (sinistral) and right-handed (dextral) forms.
Levels of Organic Structure and Isomerism
Organic compounds can be described at several structural levels, each associated with a type of isomerism:
Composition: The molecular formula.
Constitution: The connectivity of atoms (bonding patterns).
Configuration: The three-dimensional arrangement of atoms.
Conformation: The spatial arrangement due to bond rotations.
Configurational isomers (stereoisomers): Differ in configuration but are identical in constitution and composition.
Chirality in Molecules
A molecule is chiral if it is not superimposable on its mirror image. If it is superimposable, it is achiral. Chiral molecules exhibit a special type of configurational isomerism called enantiomerism.
Enantiomers are pairs of molecules that are non-superimposable mirror images of each other.
A molecule cannot be an enantiomer by itself; enantiomerism is a relationship between two molecules.
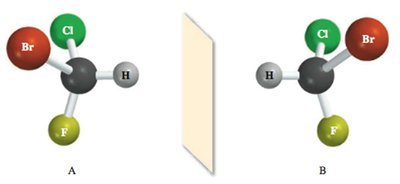
Testing for Chirality
To determine if a molecule is chiral, reflect it through a mirror and attempt to superimpose the mirror image on the original. If this is not possible, the molecule is chiral.
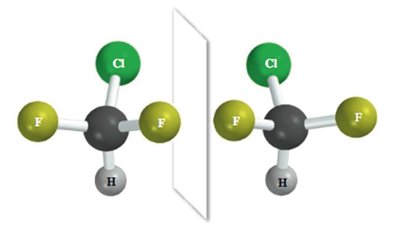
The Chirality Center
Definition and Identification
A chirality center (also called a chiral center or stereocenter) is typically a carbon atom bonded to four different groups. The presence of a single chirality center in a molecule usually makes the molecule chiral.
If a carbon atom is bonded to four different substituents, it is a chirality center.
Molecules with more than one chirality center may still be achiral if they possess certain symmetry elements.
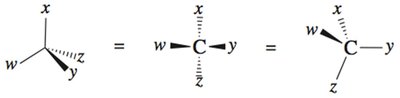
Chirality Centers in Rings
In cyclic compounds, a carbon atom can be a chirality center if the paths around the ring from that carbon are different.
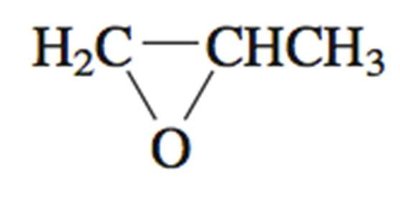
Symmetry in Achiral Structures
Elements of Symmetry
Achiral molecules often contain symmetry elements that make their mirror images superimposable:
Plane of symmetry: A plane dividing the molecule into two mirror-image halves.
Center of symmetry (inversion center): A point at the center of the molecule where any line drawn through it encounters identical elements at equal distances in opposite directions.
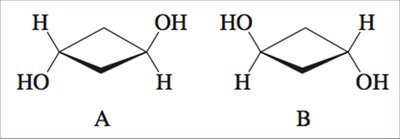
Shortcut for Identifying Chirality
If a molecule contains a plane of symmetry or a center of symmetry, it is necessarily achiral. However, generating the mirror image and attempting superimposition is the most reliable method.
Optical Activity
Rotation of Plane-Polarized Light
Chiral compounds can rotate the plane of plane-polarized light, a property known as optical activity. This rotation is measured using a polarimeter.
Dextrorotatory (+): Rotates light clockwise.
Levorotatory (–): Rotates light counterclockwise.
The direction of rotation (+ or –) is determined experimentally and is not related to the R/S configuration.
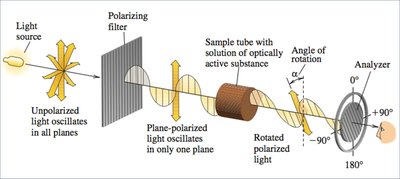
Specific Rotation and Enantiomeric Excess
The specific rotation is a standardized measure of a compound's ability to rotate plane-polarized light, accounting for concentration, path length, and temperature:
Specific rotation is calculated as:
Where is the observed rotation, is the path length in decimeters, and is the concentration in g/100 mL.
An equimolar mixture of enantiomers (racemic mixture) shows no optical activity.
Enantiomeric excess (ee):
Absolute and Relative Configuration
Definitions
Absolute configuration specifies the exact three-dimensional arrangement of atoms in a chiral molecule, often determined by X-ray crystallography. Relative configuration compares the configuration of two related compounds, provided no chirality centers are altered during conversion.
Compounds with the same relative configuration may have opposite optical rotations.
The sign of optical rotation cannot be predicted from structure alone.
Cahn–Ingold–Prelog R,S Notation
The R-S System
The R/S system assigns absolute configuration to stereocenters using a set of prioritization rules:
Assign priorities to the four groups attached to the stereocenter based on atomic number (higher atomic number = higher priority).
Orient the molecule so the group with the lowest priority is pointing away from you.
Trace a path from priority 1 → 2 → 3. If the path is clockwise, the configuration is R (rectus); if counterclockwise, it is S (sinister).
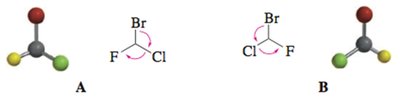
Prioritization Rules Table
Rule | Example |
|---|---|
1. Higher atomic number takes precedence over lower. | Br > Cl > O > F > H |
2. When two atoms directly attached to the chirality center are the same, compare the atoms attached to them on the basis of their atomic numbers at the first point of difference. | Ethyl (–C2H5) outranks methyl (–CH3). |

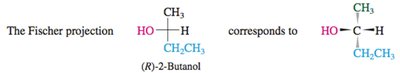
Fischer Projections
Representation and Assignment
Fischer projections are a two-dimensional representation of three-dimensional molecules, especially useful for carbohydrates and amino acids. In these projections:
Horizontal lines represent bonds coming out of the plane (toward the viewer).
Vertical lines represent bonds going behind the plane (away from the viewer).
To assign R/S, rotate the projection so the lowest priority group is on a vertical line.
Properties of Enantiomers
Physical and Chemical Properties
Pure enantiomers have identical physical properties (boiling point, melting point, density) except for their interaction with plane-polarized light and chiral environments (e.g., biological receptors).
Enantiomers may have different smells or tastes due to chiral receptors in the body.
Chirality Without a Chirality Center: Axial Chirality
Axial Chirality
Some molecules are chiral due to restricted rotation about a bond (axial chirality), even without a traditional chirality center. Examples include certain biphenyls and BINAP ligands used in asymmetric catalysis.
These enantiomers are called atropisomers.
Molecules with Multiple Chirality Centers
Multiple Stereocenters and Diastereomers
A molecule with N chirality centers can have up to stereoisomers, unless symmetry reduces the number. Stereoisomers that are not mirror images are called diastereomers.
Example: 2,3-dihydroxybutanoic acid has four possible stereoisomers, including enantiomers and diastereomers.
Meso compounds are achiral despite having chirality centers, due to internal symmetry.
Resolution of Enantiomers
Separation Techniques
Because enantiomers have identical physical properties, they are difficult to separate. Resolution involves converting enantiomers into diastereomers using a chiral agent, which can then be separated due to their differing properties.
Louis Pasteur first resolved enantiomers of tartaric acid using this principle.
Chirality Centers Other Than Carbon
Other Elements as Chirality Centers
Any atom with a tetrahedral arrangement of four different groups can be a chirality center, including silicon, phosphorus, and sulfur. Nitrogen can also be a chirality center, but rapid inversion (umbrella flipping) usually prevents isolation of enantiomers at room temperature.
Amines are generally achiral due to rapid inversion.
