 Back
BackDiels-Alder Reaction: Synthesis, Mechanism, and Analysis
Study Guide - Smart Notes

Diels-Alder Reaction and Polycyclic Aromatic Compound Synthesis
Introduction to Hydrocarbons and Dienes
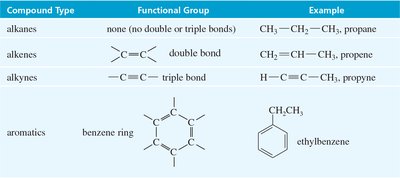
Hydrocarbons are organic molecules composed solely of carbon and hydrogen. They are classified based on the types of bonds between carbon atoms, which influences their chemical reactivity and physical properties. Dienes, a subclass of hydrocarbons, contain two double bonds and play a crucial role in cycloaddition reactions such as the Diels-Alder reaction.
Alkanes: Only single bonds between carbons.
Alkenes: Contain at least one carbon–carbon double bond.
Alkynes: Contain at least one carbon–carbon triple bond.
Aromatics: Contain benzene rings with delocalized π electrons.
Classification of Dienes
Conjugated Dienes: Double bonds separated by a single bond (e.g., 1,3-butadiene).
Isolated Dienes: Double bonds separated by two or more single bonds (e.g., 1,4-pentadiene).
Cumulated Dienes: Double bonds on adjacent carbons (e.g., allene).
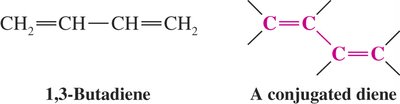
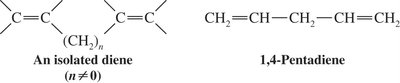
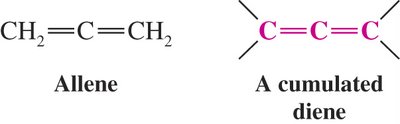
Diels-Alder Reaction: Mechanism and Features
The Diels-Alder reaction is a [4+2] cycloaddition between a conjugated diene and a dienophile (an alkene or alkyne with electron-withdrawing groups), resulting in the formation of a six-membered ring. This reaction is concerted, meaning all bond-making and bond-breaking events occur simultaneously.
Concerted Mechanism: Involves the movement of three π electron pairs in a single step.
Diene: Provides four π electrons.
Dienophile: Provides two π electrons, often activated by electron-withdrawing groups.
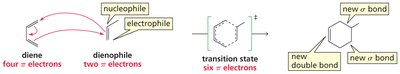
Factors Affecting Diels-Alder Reaction
Electron-Withdrawing Groups (EWG): On the dienophile, these groups increase reactivity by making the alkene more electrophilic.
Electron-Donating Groups (EDG): On the diene, these groups increase nucleophilicity and reaction rate.
Conformational Requirement: The diene must be in the s-cis conformation to react efficiently.
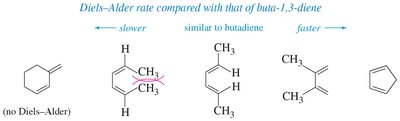
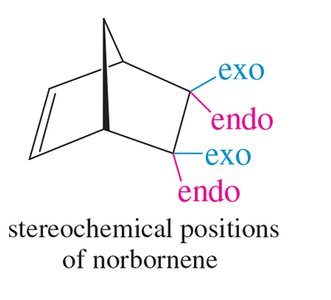
Rules and Stereochemistry of the Diels-Alder Reaction
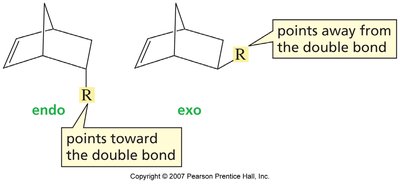
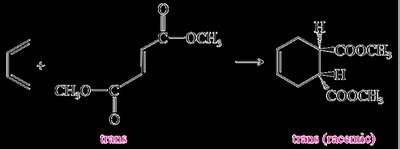
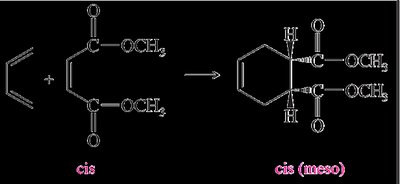
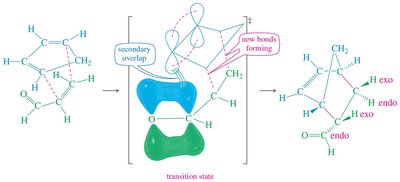
The Diels-Alder reaction is both regioselective and stereospecific. The stereochemistry of the dienophile is retained in the product, and the reaction typically favors the formation of the endo product due to secondary orbital interactions (Alder-Endo Rule).
s-cis Requirement: Only dienes in the s-cis conformation can participate in the reaction.
Stereospecificity: The configuration (cis or trans) of the dienophile is preserved in the product.
Endo Rule: Electron-withdrawing substituents on the dienophile prefer the endo position in the bicyclic product due to favorable orbital overlap.
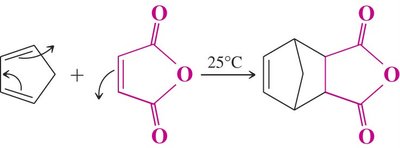
Experiment: Synthesis of a Bridged Polycyclic Aromatic Compound
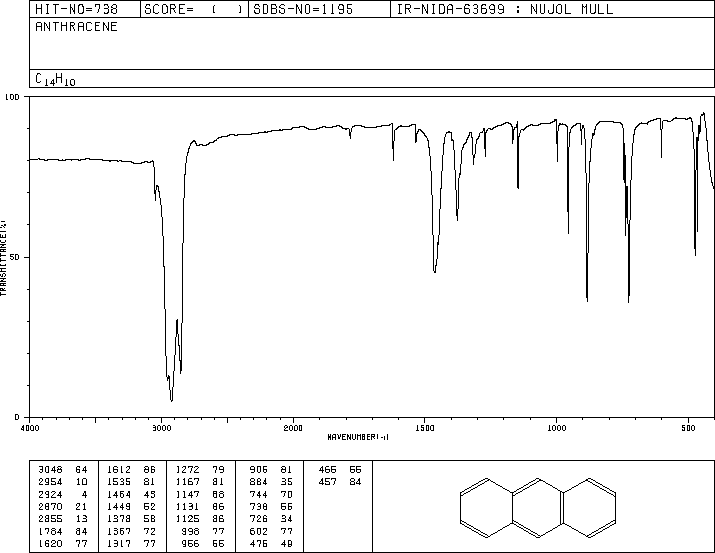
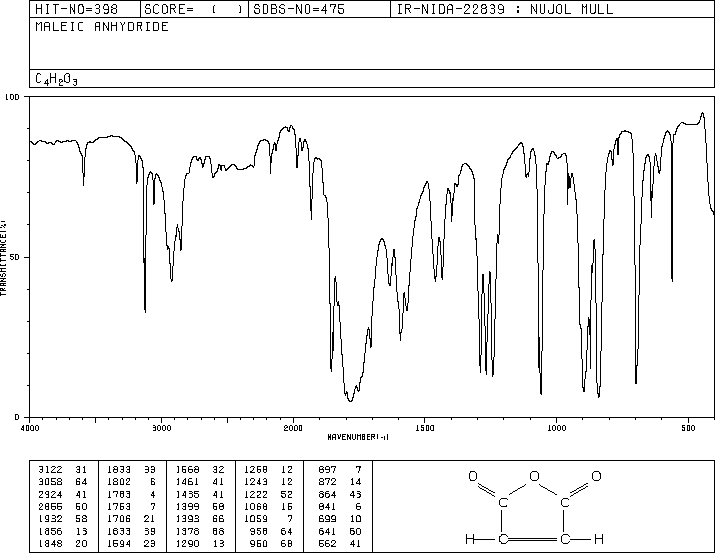
In this experiment, anthracene (a polycyclic aromatic hydrocarbon) reacts with maleic anhydride (a highly activated dienophile) in xylene to form a bridged polycyclic product via the Diels-Alder reaction. The reaction is performed under reflux, and the product is isolated by vacuum filtration and analyzed by melting point and IR spectroscopy.
Anthracene: Acts as the diene, reacting through its central ring.
Maleic Anhydride: Acts as the dienophile, activated by two carbonyl groups.
Xylene: Used as a high-boiling solvent to facilitate the reaction and crystallization.
Product: A bridged polycyclic anhydride, stable due to aromaticity in the outer rings.
Experimental Setup and Safety
Wear full PPE: goggles, apron, gloves, and closed shoes.
Work in a fume hood due to the toxicity and volatility of reagents.
Dispose of all chemical waste according to lab protocols.



Analysis of Reaction Products
Product identity and purity are confirmed by melting point determination and IR spectroscopy. The IR spectra of anthracene, maleic anhydride, and the Diels-Alder adduct are compared to verify the formation of the new compound.
Anthracene IR: Shows characteristic aromatic C-H and C=C stretches.
Maleic Anhydride IR: Shows strong C=O stretches and anhydride fingerprint region.
Product IR: Should show new absorptions corresponding to the anhydride and loss of alkene signals.
Summary Table: Diene Classification
Type of Diene | Structure | Example |
|---|---|---|
Conjugated | Double bonds separated by one single bond | 1,3-Butadiene |
Isolated | Double bonds separated by two or more single bonds | 1,4-Pentadiene |
Cumulated | Double bonds on adjacent carbons | Allene |
Key Equations
Diels-Alder General Equation:
Example (Cyclopentadiene + Maleic Anhydride):
Additional info:
The Diels-Alder reaction is a cornerstone of synthetic organic chemistry, allowing for the efficient construction of six-membered rings with high regio- and stereoselectivity.
Understanding the IR spectra of reactants and products is essential for confirming the success of the reaction and the purity of the isolated compound.
