 Back
BackElectrophilic and Nucleophilic Aromatic Substitution: Reactions and Mechanisms
Study Guide - Smart Notes

Reactions of Aromatic Compounds
Electrophilic Aromatic Substitution (EAS)
Electrophilic aromatic substitution is a fundamental reaction in organic chemistry, where an aromatic ring reacts with an electrophile, resulting in the substitution of a hydrogen atom. The aromatic ring's stability is preserved throughout the process, making these reactions crucial for the functionalization of benzene and its derivatives.
Halogenation
Halogenation introduces a halogen atom (Cl or Br) onto the benzene ring using a halogen and a Lewis acid catalyst. The reaction proceeds via the generation of a halonium ion, which acts as the electrophile.
Chlorination: Benzene reacts with Cl2 and FeCl3 to form chlorobenzene.
Bromination: Benzene reacts with Br2 and FeBr3 to form bromobenzene.
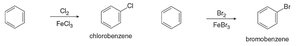
Note: Iodination and fluorination are not synthetically useful due to reactivity issues.
Nitration and Sulfonation
Nitration and sulfonation are important methods for introducing nitro and sulfonic acid groups onto aromatic rings. These reactions use strong acids to generate the electrophiles.
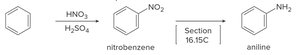
Nitration: Benzene reacts with HNO3 and H2SO4 to form nitrobenzene.
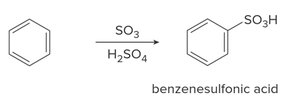
Sulfonation: Benzene reacts with SO3 and H2SO4 to form benzenesulfonic acid.
Friedel–Crafts Alkylation
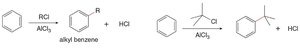
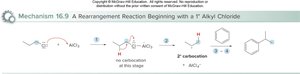
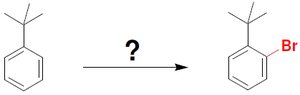
Friedel–Crafts alkylation introduces an alkyl group onto the benzene ring using an alkyl halide and AlCl3 as a catalyst. The reaction can lead to rearrangements and polyalkylation.
Mechanism: Alkyl halide reacts with AlCl3 to generate a carbocation, which then reacts with benzene.
Rearrangements: Carbocation rearrangements can occur, even if not initially formed.
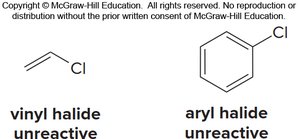
Limitations: Vinyl and aryl halides do not react; polyalkylation is possible.
Friedel–Crafts Acylation
Friedel–Crafts acylation introduces an acyl group onto the benzene ring using an acid chloride and AlCl3. The product is a ketone, and the reaction avoids polyacylation.
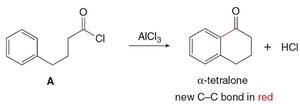
Mechanism: Acid chloride reacts with AlCl3 to form an acylium ion, which then reacts with benzene.
Example: Formation of α-tetralone from a substituted acid chloride.
Substituent Effects on Aromatic Reactivity
Substituents on the benzene ring influence both the rate and the site of electrophilic substitution. These effects are classified as electron-withdrawing or electron-donating, and they determine whether new substituents are directed to the ortho, meta, or para positions.
Inductive and Resonance Effects
Electron-withdrawing groups: Decrease electron density, typically meta-directing.
Electron-donating groups: Increase electron density, typically ortho/para-directing.

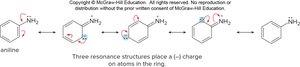
Resonance Effects
Amines: Place negative charge on ring atoms, activating the ring.
Phenols: Similar resonance activation as amines.
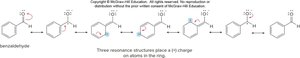
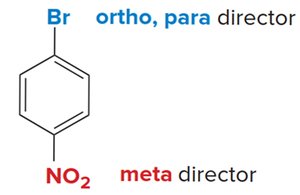
Site-Selectivity and Directing Effects
The position at which new substituents are introduced depends on the nature of existing groups. Ortho/para directors are typically electron-donating, while meta directors are electron-withdrawing.
Ortho/para directors: Alkyl, hydroxyl, amino groups.
Meta directors: Nitro, carbonyl, and other electron-withdrawing groups.
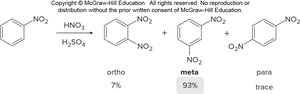
Summary Table: Reactivity and Directing Effects
Substituent | Directing Effect | Activation/Deactivation |
|---|---|---|
Alkyl | Ortho/Para | Activating |
OH, NH2 | Ortho/Para | Strongly Activating |
NO2, COOH | Meta | Deactivating |
Halogen | Ortho/Para | Deactivating |
Special Cases in Aromatic Substitution
Strong electron-donating groups (OH, NH2, OR, NHR, NR2) can cause polyhalogenation. Strong electron-withdrawing groups (meta directors) deactivate the ring, preventing Friedel–Crafts reactions.
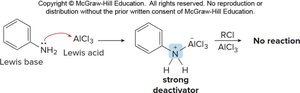
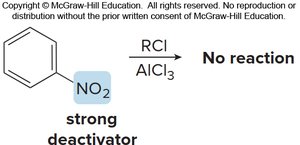
Disubstituted Benzenes
When two substituents are present, their combined effects determine the site of further substitution. The more activating group usually dominates.
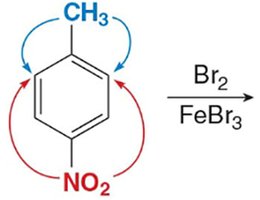
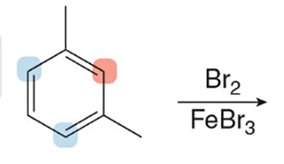
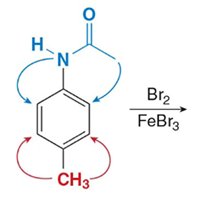
Synthesis of Benzene Derivatives
Complex benzene derivatives can be synthesized by sequential substitution reactions, considering the directing effects of each substituent.
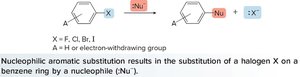
Nucleophilic Aromatic Substitution (NAS)
Mechanism and Requirements
Nucleophilic aromatic substitution occurs when a nucleophile replaces a leaving group (usually a halogen) on an aromatic ring. The ring must be activated by electron-withdrawing groups to stabilize the intermediate.
Addition-Elimination: The nucleophile adds to the ring, forming a Meisenheimer complex, followed by elimination of the leaving group.
Elimination-Addition: The leaving group is eliminated first, followed by nucleophilic addition.
Reactions of Alkyl Benzenes
Halogenation of Alkyl Benzenes
Alkyl benzenes can undergo halogenation at the benzylic position, which is highly reactive due to resonance stabilization.
Oxidation of Alkyl Benzenes
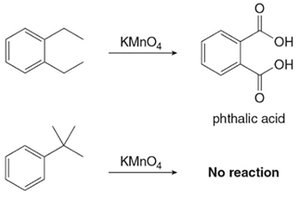
Alkyl benzenes with at least one benzylic C–H bond are oxidized by KMnO4 to benzoic acid derivatives. If no benzylic hydrogen is present, no reaction occurs.
Reduction of Acyl Benzenes
Acyl benzenes can be reduced to alkyl benzenes using catalytic hydrogenation (Clemmensen or Wolff-Kishner reduction).

Reduction of Nitro Benzenes
Nitro groups on benzene can be reduced to amines, providing a route to aniline derivatives.
Target Synthesis
Target synthesis involves planning a sequence of reactions to obtain a desired aromatic compound, considering the reactivity and directing effects of substituents.
Additional info: The notes cover all major aspects of Chapter 16: Electrophilic and Nucleophilic Aromatic Substitution, including mechanisms, substituent effects, and synthetic applications. Tables and images are included only when directly relevant to the explanation.
