 Back
BackExam 2 Review – Organic Chemistry Nomenclature, Isomerism, and Physical Properties
Study Guide - Smart Notes

Q10. What is the relationship between the compounds shown below?
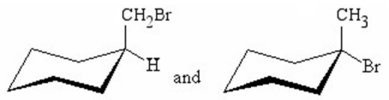
Background
Topic: Stereochemistry – Isomerism
This question tests your understanding of the different types of isomerism in organic chemistry, specifically how to distinguish between constitutional isomers, enantiomers, diastereomers, meso compounds, and identical compounds. You are asked to analyze two cyclohexane derivatives and determine their relationship.
Key Terms and Concepts:
Constitutional isomers: Compounds with the same molecular formula but different connectivity of atoms.
Enantiomers: Non-superimposable mirror images (chiral pairs).
Diastereomers: Stereoisomers that are not mirror images.
Meso compounds: Compounds with stereocenters that are achiral due to an internal plane of symmetry.
Identical compounds: The same compound, possibly drawn differently.
Step-by-Step Guidance
Carefully examine the two cyclohexane chair structures. Identify the substituents on each ring and their positions (axial/equatorial, up/down).
Assign priorities to the substituents on each stereocenter using the Cahn-Ingold-Prelog rules. Determine the configuration (R or S) for each stereocenter in both molecules.
Check if the two molecules are mirror images of each other (enantiomers), non-mirror image stereoisomers (diastereomers), or if they are the same compound drawn differently (identical compounds).
Consider whether the compounds could be meso (achiral with stereocenters) or constitutional isomers (different connectivity).
Try solving on your own before revealing the answer!
Final Answer: Diastereomers
The two compounds are diastereomers. They are stereoisomers that are not mirror images of each other. This is because the substituents are arranged differently on the cyclohexane ring, leading to different configurations at the stereocenters.
