 Back
BackIodination of 4’-Hydroxyacetophenone: Electrophilic Aromatic Substitution and Green Chemistry
Study Guide - Smart Notes

Iodination of 4’-Hydroxyacetophenone
Introduction to Aromatic Compounds
Aromatic compounds are a class of cyclic polyenes characterized by planar rings with alternating single and double bonds, resulting in unique stability and reactivity. These compounds, such as benzene, are resistant to addition reactions and instead undergo electrophilic aromatic substitution (EAS). Aromatic compounds are widely used in solvents, pharmaceuticals, plastics, and dyes.
Definition: Aromatic compounds contain conjugated pi electron systems in a cyclic, planar structure, following Hückel’s rule (4n+2 pi electrons).
Properties: High stability, non-polar, hydrophobic, high carbon-hydrogen ratio, sooty yellow flame, undergo EAS and nucleophilic aromatic substitution.

Electrophilic Aromatic Substitution (EAS)
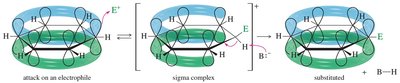
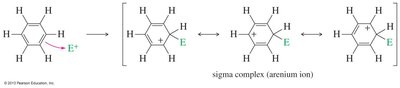
EAS is the primary reaction mechanism for aromatic compounds, where an electrophile replaces a hydrogen atom on the aromatic ring. The reaction proceeds via a two-step mechanism: formation of a resonance-stabilized sigma complex (arenium ion), followed by deprotonation to restore aromaticity.
Step 1: The aromatic pi electrons attack a strong electrophile, forming a sigma complex (arenium ion).
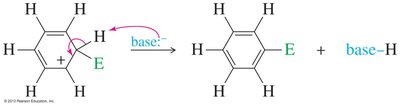
Step 2: Loss of a proton from the sigma complex restores aromaticity and yields the substituted product.
Reactivity and Regioselectivity in EAS
The rate and position of substitution in EAS are influenced by substituents already present on the aromatic ring. Electron-donating groups (EDGs) increase reactivity and direct new substituents to ortho and para positions, while electron-withdrawing groups (EWGs) decrease reactivity and direct new substituents to the meta position.
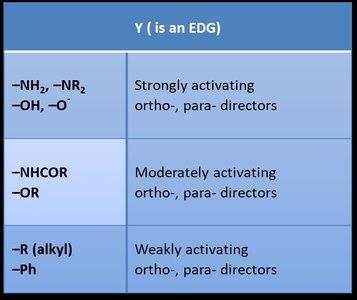
EDGs: Increase electron density, activate the ring, ortho/para directors.
EWGs: Decrease electron density, deactivate the ring, meta directors.
Y (is an EDG) | Effect |
|---|---|
–NH2, –NR2, –OH, –O– | Strongly activating, ortho-, para- directors |
–NHCOR, –OR | Moderately activating, ortho-, para- directors |
–R (alkyl), –Ph | Weakly activating, ortho-, para- directors |
Y (is an EWG) | Effect |
|---|---|
–Halide (F, Cl, Br, I) | Weakly deactivating, ortho-, para- directors |
–COOR, –COR, –CHO, –COOH, –SO3H, –CN | Moderately deactivating, meta directors |
–CF3, –CCl3, –NO2, –⊕NR3 | Strongly deactivating, meta directors |

Green Chemistry: Iodination of 4’-Hydroxyacetophenone
Overview and Purpose
This experiment demonstrates the iodination of 4’-hydroxyacetophenone via EAS using green chemistry principles. Traditional iodination methods use hazardous oxidants like nitric acid, but this procedure generates the electrophilic iodine species in situ using sodium iodide and household bleach (sodium hypochlorite) in aqueous ethanol, minimizing hazardous waste and exposure.
Reaction:
Green Chemistry: Avoids toxic oxidants, uses safer reagents and solvents, and is more selective for mono-iodination.
Regioselectivity in Iodination of 4’-Hydroxyacetophenone
4’-Hydroxyacetophenone contains both an activating group (hydroxyl, –OH) and a deactivating group (acetyl, –COCH3). The –OH group is a strong ortho/para director, while the acetyl group is a meta director. The major product is determined by the combined electronic and steric effects, with the ortho position to the –OH group (and meta to the acetyl group) being most favored.
Major Product: Ortho-iodo-4’-hydroxyacetophenone
Minor Product: Para-iodo-4’-hydroxyacetophenone (less favored due to steric hindrance)
Experimental Procedure
Reaction Setup
The reaction is performed in a large test tube with a magnetic stir bar, clamped over a hot/stir plate. The solid 4’-hydroxyacetophenone is dissolved in ethanol, followed by addition of sodium iodide. The mixture is cooled in an ice/water bath before bleach is added dropwise to generate the electrophilic iodine species.


Reaction Workup and Isolation
After the reaction, sodium thiosulfate is added to neutralize excess oxidants and iodine. The mixture is acidified with hydrochloric acid to precipitate the aryl iodide product. The solid is isolated by vacuum filtration using a Hirsch funnel and filtration flask, washed with cold water and ethanol, and dried.

Safety and Waste Disposal
Wear splash-proof goggles, chemically resistant aprons, and gloves at all times.
Handle all chemicals in a fume hood; many reagents are irritants or corrosive.
Dispose of liquid and solid waste in designated containers; do not pour chemicals down the drain.
Follow all lab protocols for cleanliness, equipment return, and post-lab cleanup.
Summary Table: Key Steps in the Experiment
Step | Description |
|---|---|
1 | Dissolve 4’-hydroxyacetophenone in ethanol, add sodium iodide |
2 | Cool in ice/water bath, add bleach dropwise |
3 | Add sodium thiosulfate, acidify with HCl |
4 | Isolate product by vacuum filtration, wash and dry |
Additional info:
Green chemistry principles are emphasized to reduce hazardous waste and improve safety.
Understanding the directing effects of substituents is crucial for predicting the major product in EAS reactions.
