 Back
BackLipids: Structure, Classification, and Biological Roles
Study Guide - Smart Notes

Unit 8: Lipids
Introduction to Lipids
Lipids are a diverse group of naturally occurring compounds characterized by their solubility in nonpolar solvents and insolubility in water. Unlike other biomolecules, lipids are defined by their physical properties rather than a specific functional group. They play crucial roles in biological systems, including energy storage, cell membrane structure, and signaling.
Key Property: High content of C–C and C–H bonds, making them hydrophobic.
Biological Importance: Components of cell membranes, fat-soluble vitamins, and steroid hormones.
Classification of Lipids
Complex Lipids
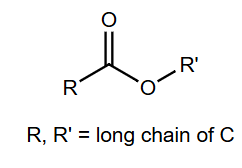
Complex lipids are hydrolyzable molecules, typically esters of long-chain carboxylic acids (fatty acids). They can be further divided into waxes, triglycerides, and phospholipids.
Waxes: Esters of long-chain fatty acids with long-chain alcohols. Highly hydrophobic and have high melting points.
Glycerides: Fatty acid esters of glycerol. The most common are triglycerides (triacylglycerols), where all three hydroxyl groups of glycerol are esterified by fatty acids.
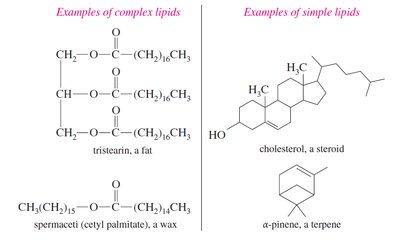
Triglycerides: Fats and Oils
Triglycerides are classified as fats if solid at room temperature and oils if liquid. Their physical state depends on the degree of saturation and the length of their fatty acid chains.
Fats: Mostly saturated fatty acids, solid at room temperature (e.g., beef tallow, lard).
Oils: Mostly unsaturated fatty acids, liquid at room temperature (e.g., corn oil, fish oil).
Energy Storage: Fats provide more than twice the energy per gram compared to carbohydrates.
Fatty Acids: Structure and Properties
Fatty acids are long, unbranched carboxylic acids (12–20 carbons), usually with even numbers of carbons. They can be saturated (no double bonds) or unsaturated (one or more double bonds).
Name | Carbons | Structure | Melting Point (°C) |
|---|---|---|---|
Lauric acid | 12 | CH3(CH2)10COOH | 44 |
Myristic acid | 14 | CH3(CH2)12COOH | 59 |
Palmitic acid | 16 | CH3(CH2)14COOH | 64 |
Stearic acid | 18 | CH3(CH2)16COOH | 70 |
Arachidic acid | 20 | CH3(CH2)18COOH | 77 |
Oleic acid | 18 | CH3(CH2)7CH=CH(CH2)7COOH | 4 |
Linoleic acid | 18 | CH3(CH2)4CH=CHCH2CH=CH(CH2)7COOH | -5 |
Linolenic acid | 18 | CH3CH2CH=CHCH2CH=CHCH2CH=CH(CH2)7COOH | -11 |
Eicosanoic acid | 20 | CH3(CH2)16COOH | -49 |
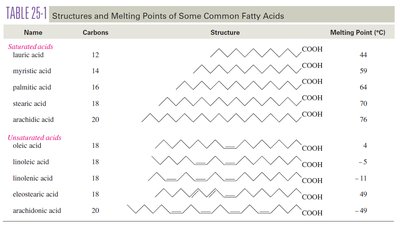
Effect of Unsaturation on Melting Point
The presence of cis double bonds introduces kinks in the fatty acid chains, preventing tight packing and lowering the melting point. For example, stearic acid (saturated, mp 70°C) vs. oleic acid (one cis double bond, mp 4°C).
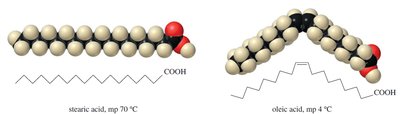
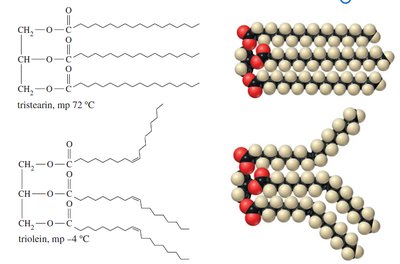
Packing of Triglycerides
Saturated triglycerides (e.g., tristearin) pack efficiently and are solid at room temperature, while unsaturated triglycerides (e.g., triolein) are kinked and remain liquid.
Omega Fatty Acids
Omega fatty acids are classified by the position of the first double bond from the methyl (omega) end. Linoleic acid (omega-6) and linolenic acid (omega-3) are essential fatty acids for mammals, as they cannot be synthesized and must be obtained from the diet. Omega-3 fatty acids are important for heart health.

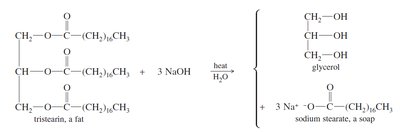
Transesterification and Saponification
Transesterification is used to convert fats and oils into biodiesel (fatty acid methyl esters) using methanol and a base catalyst. Saponification is the base-catalyzed hydrolysis of triglycerides to produce soap (salts of fatty acids) and glycerol.
Transesterification: Produces biodiesel, which is more volatile and suitable for diesel engines.
Saponification: Produces soap, which forms micelles in water to emulsify grease.
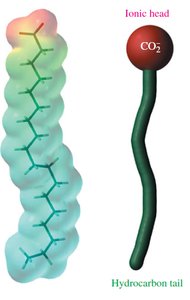
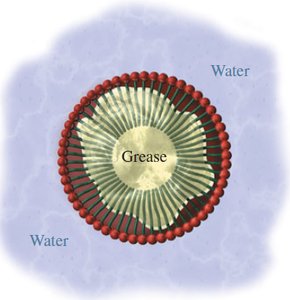
Soap Structure and Micelles
Soaps are sodium or potassium salts of fatty acids. The carboxylate group is hydrophilic, while the hydrocarbon tail is hydrophobic. In water, soaps form micelles, with hydrophilic heads on the outside and hydrophobic tails inside, trapping grease.
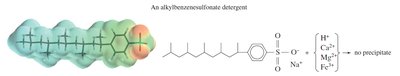
Detergents
Synthetic detergents use functional groups other than carboxylate to avoid precipitation in hard water. Their salt products remain soluble, preventing the formation of scum.
Phospholipids
Phospholipids are major components of cell membranes. They contain a phosphate group, often with an additional alcohol (e.g., choline or ethanolamine). Phospholipids can form micelles or bilayers, the latter being the structural basis of biological membranes.
Phosphatidic acids: Simplest phosphoglycerides, with two fatty acids and one phosphate group on glycerol.
Lecithins and Cephalins: Contain choline or ethanolamine, respectively.

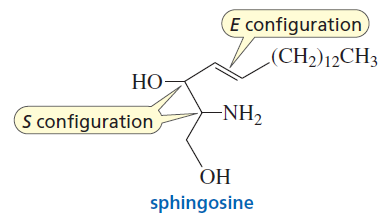
Sphingolipids
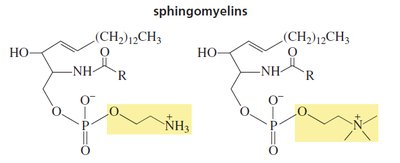
Sphingolipids are membrane lipids containing the amino alcohol sphingosine. The amino group forms an amide with a fatty acid. Sphingomyelins are the most common sphingolipids, with a phosphate group attached to the primary alcohol of sphingosine.
Simple Lipids
Simple lipids are not easily hydrolyzed and include steroids, prostaglandins, and terpenes.
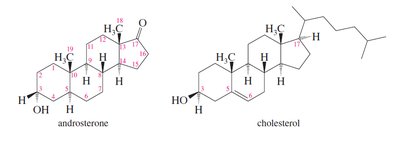
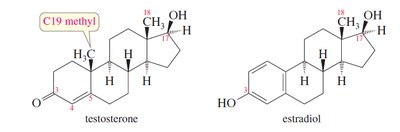
Steroids: Polycyclic molecules based on the androstane ring system. Examples include cholesterol, testosterone, and estradiol.
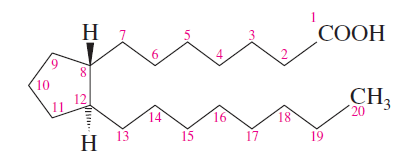
Prostaglandins: Fatty acid derivatives with a cyclopentane ring, involved in diverse physiological processes.
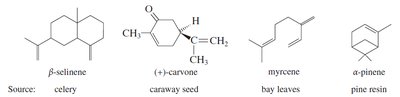
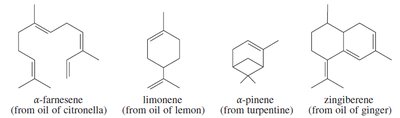
Terpenes: Compounds built from isoprene units, found in essential oils of plants.

Steroids
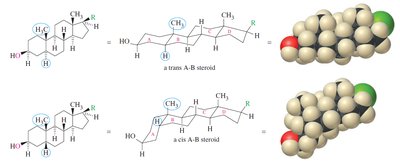
Steroids have a tetracyclic ring system and are classified by their ring junctions (cis or trans). Cholesterol is a key membrane component and precursor to other steroids. Testosterone and estradiol are sex hormones, differing by the presence of an aromatic ring in estradiol.
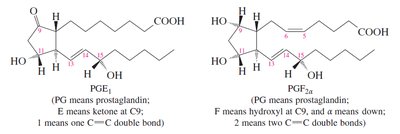
Prostaglandins
Prostaglandins are potent regulators of physiological functions, including inflammation, blood pressure, and smooth muscle activity. They have a cyclopentane ring and two long side chains, with various functional groups determining their classification (E or F series).
Terpenes
Terpenes are constructed from isoprene units (C5H8). They are found in essential oils and have diverse structures and functions. The isoprene rule describes their assembly in a head-to-tail fashion.
Additional info: This summary includes expanded academic context and logical grouping of fragmented topics for clarity and completeness.
