 Back
BackNerolin Synthesis and Nucleophilic Substitution: SN2 Mechanism and Laboratory Practice
Study Guide - Smart Notes

Nucleophilic Substitution Reactions: SN1 and SN2 Mechanisms
Overview of Nucleophilic Substitution
Nucleophilic substitution reactions are fundamental processes in organic chemistry where a nucleophile replaces a leaving group attached to an sp3 hybridized carbon. These reactions are classified as either unimolecular (SN1) or bimolecular (SN2) based on their mechanisms.
SN1 Mechanism: Involves a two-step process with carbocation intermediate formation. Favored by tertiary substrates and polar protic solvents.
SN2 Mechanism: Involves a single concerted step where the nucleophile attacks the substrate as the leaving group departs. Favored by primary substrates and polar aprotic solvents.
Preparation of Alkyl Halides from Alcohols
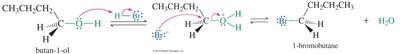
Alcohols can be converted to alkyl halides via nucleophilic substitution with hydrogen halides. The hydroxyl group is a poor leaving group, but protonation converts it into a better leaving group (water), facilitating substitution.
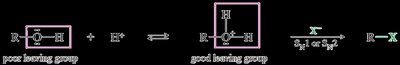
Protonation: The alcohol is protonated by acid, forming an oxonium ion (good leaving group).
Substitution: The halide ion replaces water, forming the alkyl halide.
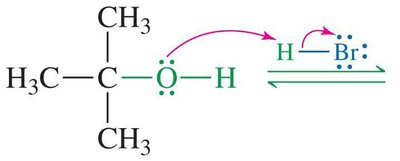
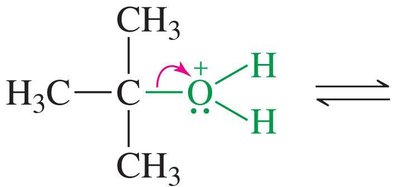
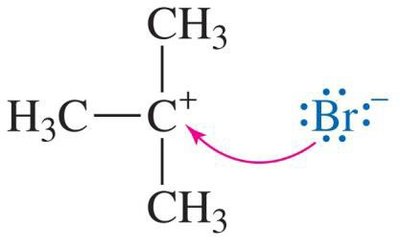
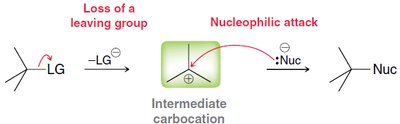
SN1 Mechanism: Example and Steps
The SN1 mechanism is typical for tertiary alcohols due to the stability of the resulting carbocation. The reaction proceeds via three main steps:
Protonation of the alcohol
Loss of water to form a carbocation
Nucleophilic attack by the halide ion
SN2 Mechanism: Example and Steps
The SN2 mechanism is favored by primary alcohols and involves a concerted, one-step process. The nucleophile attacks the substrate from the opposite side of the leaving group, resulting in inversion of configuration.
Key Features: Bimolecular, single transition state, no carbocation intermediate, stereospecific inversion.
General Mechanisms of SN1 and SN2
SN2: Nucleophile attacks as leaving group departs.
SN1: Leaving group departs first, forming a carbocation, then nucleophile attacks.
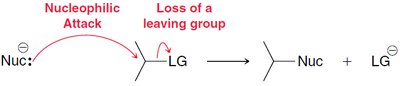
Williamson Ether Synthesis and Nerolin Preparation
Williamson Ether Synthesis
The Williamson ether synthesis is a classic method for preparing ethers by reacting an alkoxide ion with a primary alkyl halide under SN2 conditions. The reaction is highly dependent on the structure of the alkyl halide, the strength of the nucleophile, and the solvent used.
Alkoxide Ion: Generated by deprotonating an alcohol with a strong base.
Substrate: Primary alkyl halides are preferred for high yields.
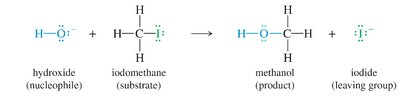
Mechanistic Steps
Proton transfer to generate the alkoxide ion
Nucleophilic attack on the alkyl halide

Acidity of Alcohols and Phenols
Comparative Acidity
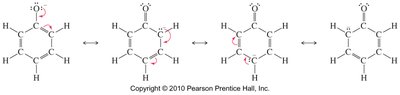
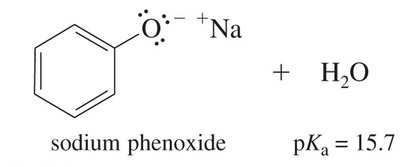
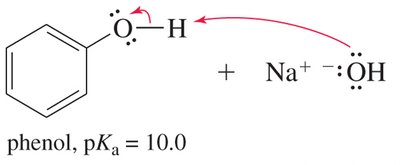
Alcohols are weak acids, but phenols are significantly more acidic due to resonance stabilization of the phenoxide ion. The negative charge on the oxygen atom in phenoxide is delocalized over the aromatic ring, increasing stability and acidity.
Alcohols: Weakly acidic, pKa typically around 16-18.
Phenols: Much more acidic, pKa around 10, due to resonance stabilization.

Laboratory Techniques: Reflux and Filtration
Reflux Setup
Reflux is a technique used to heat chemical reactions for extended periods without loss of solvent. Proper assembly of the reflux apparatus is essential for safety and efficiency.
Lightly grease the ground glass joint of the condenser.
Attach connectors and O-rings as shown.

Connect water hoses to the condenser (water in at the bottom, water out at the top).
Clamp the round bottom flask and condenser securely.

Vacuum Filtration Setup
Vacuum filtration is used to separate solid products from reaction mixtures efficiently. The Hirsch funnel and side-arm flask are essential components.
Clamp the Hirsch funnel and filtration flask.
Connect the flask to a vacuum source (water aspirator).
Ensure filter paper is properly placed and wetted.

Infrared Spectroscopy (IR) of Reactants
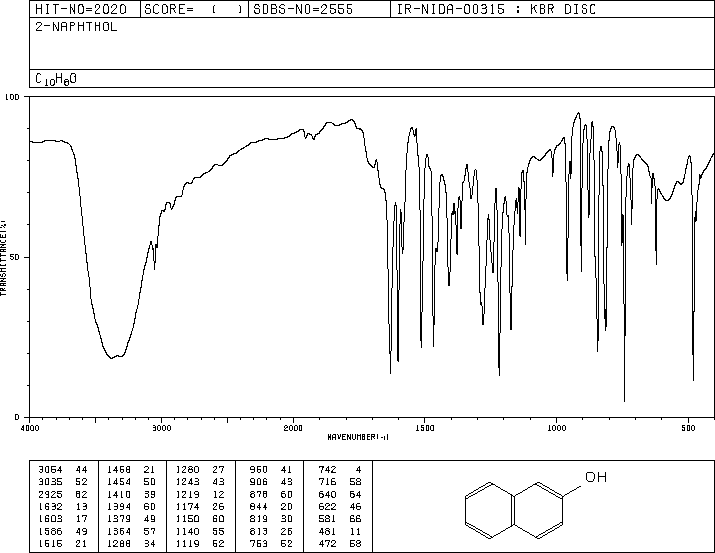
IR Spectrum of 2-Naphthol
2-Naphthol shows characteristic IR absorptions for O-H stretching (broad, around 3200-3600 cm-1), aromatic C-H stretching, and C-O stretching (1000-1300 cm-1).
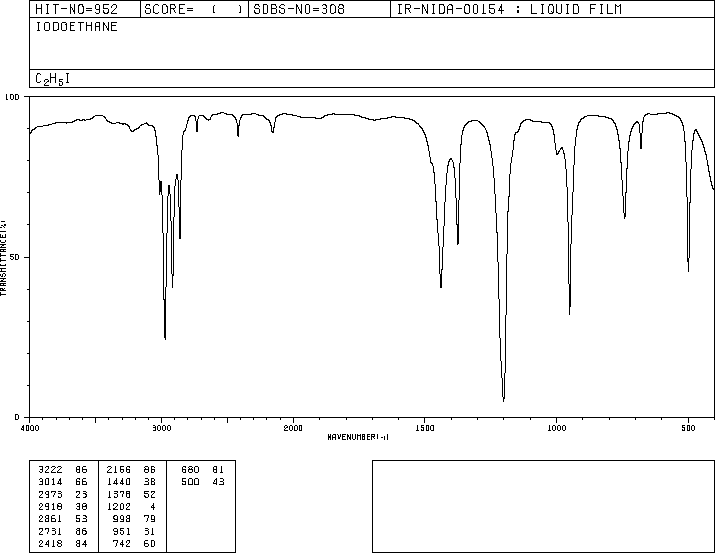
IR Spectrum of Iodoethane
Iodoethane displays C-H stretching (around 2900-3000 cm-1) and C-I stretching (500-600 cm-1).
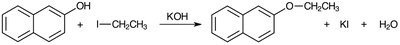
Synthesis of Nerolin (2-Ethoxynaphthalene)
Reaction Equation and Mechanism
Nerolin is synthesized via the Williamson ether synthesis, where 2-naphthol is deprotonated by potassium hydroxide to form the naphthoxide ion, which then attacks ethyl iodide in an SN2 reaction to yield 2-ethoxynaphthalene (Nerolin).
Base: Potassium hydroxide (KOH) is used due to its solubility in ethanol.
Solvent: Ethanol is chosen for its ability to dissolve both reactants and base.
Product Isolation: Nerolin precipitates upon addition of ice-cold water and is purified by recrystallization.
Summary Table: SN1 vs SN2 Mechanisms
Feature | SN1 | SN2 |
|---|---|---|
Mechanism | Two-step (carbocation intermediate) | One-step (concerted) |
Substrate Preference | Tertiary > Secondary > Primary | Methyl > Primary > Secondary |
Stereochemistry | Racemization | Inversion of configuration |
Rate Law | Rate = k[substrate] | Rate = k[substrate][nucleophile] |
Solvent | Polar protic | Polar aprotic |
Key Safety and Laboratory Practices
Always wear appropriate PPE: goggles, apron, gloves, and closed-toe shoes.
Work in a fume hood when handling volatile or toxic chemicals.
Dispose of chemical waste according to laboratory protocols.
Clean all glassware and work areas after completing experiments.
