 Back
BackNomenclature, Structure, and Conformational Analysis of Alkanes and Cycloalkanes
Study Guide - Smart Notes

Chapter 3 – Introduction to Organic Compounds
Nomenclature of Straight-Chain Alkanes
Straight-chain alkanes are saturated hydrocarbons containing only carbon and hydrogen atoms, with all carbon-carbon bonds being single bonds. The systematic naming of alkanes follows the IUPAC rules, which assign names based on the number of carbon atoms in the chain.
Hydrocarbon: A compound made only of hydrogen and carbon.
Alkane: A hydrocarbon with only C–C single bonds.
Straight-chain vs. Branched: Straight-chain alkanes have a continuous chain of carbon atoms, while branched alkanes have side chains.
Constitutional Isomers: Compounds with the same molecular formula but different bonding arrangements.
IUPAC System: The systematic method for naming organic compounds, preferred over common names.
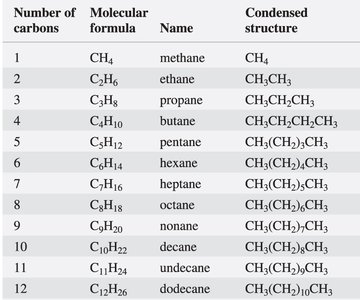
Example: The table below shows the names, molecular formulas, and condensed structures for straight-chain alkanes from methane to dodecane.
Number of carbons | Molecular formula | Name | Condensed structure |
|---|---|---|---|
1 | CH4 | methane | CH4 |
2 | C2H6 | ethane | CH3CH3 |
3 | C3H8 | propane | CH3CH2CH3 |
4 | C4H10 | butane | CH3CH2CH2CH3 |
5 | C5H12 | pentane | CH3(CH2)3CH3 |
6 | C6H14 | hexane | CH3(CH2)4CH3 |
7 | C7H16 | heptane | CH3(CH2)5CH3 |
8 | C8H18 | octane | CH3(CH2)6CH3 |
9 | C9H20 | nonane | CH3(CH2)7CH3 |
10 | C10H22 | decane | CH3(CH2)8CH3 |
11 | C11H24 | undecane | CH3(CH2)9CH3 |
12 | C12H26 | dodecane | CH3(CH2)10CH3 |
Functional Groups and Classification
Functional groups are specific groups of atoms within molecules that are responsible for the characteristic chemical reactions of those molecules. Alkyl halides, alcohols, and amines are classified based on the type of carbon atom to which the functional group is attached.
Alkyl Halide: Compound with a C–X bond (X = F, Cl, Br, I).
Alcohol: Compound with a C–OH bond.
Amine: Compound with a C–N bond.
Classification: Primary, secondary, tertiary, and quaternary, depending on the number of carbon atoms attached to the functional group.
Example: A primary alcohol is attached to one carbon, a secondary to two, and a tertiary to three.
Conformational Analysis of Alkanes
Rotation Around C–C σ Bonds
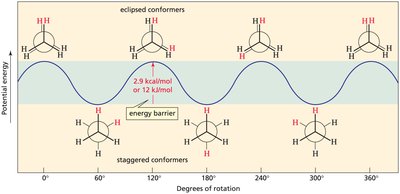
Alkanes can rotate around their carbon-carbon single (σ) bonds, leading to different spatial arrangements called conformers. These conformers are interconverted by rotation and are classified as staggered or eclipsed based on the relative positions of atoms.
Conformational Isomers (Conformers): Molecules with the same connectivity but different spatial arrangements due to rotation about single bonds.
Newman Projection: A way to visualize the spatial arrangement of atoms around a bond by looking straight down the bond axis.
Staggered Conformer: Atoms are positioned so that bonds are as far apart as possible, minimizing repulsion.
Eclipsed Conformer: Atoms are aligned, maximizing repulsion and making the molecule less stable.


Energy Differences Between Conformers
The staggered conformer is more stable than the eclipsed conformer due to reduced electron repulsion and the stabilizing effect of hyperconjugation. The energy barrier between these conformers is typically around 2.9 kcal/mol for ethane.
Hyperconjugation: Delocalization of electrons from filled bonding orbitals to empty antibonding orbitals, stabilizing the staggered form.
Potential Energy Diagram: Shows the energy changes as the molecule rotates about the C–C bond.

Conformers of Butane
Butane exhibits several conformers due to rotation about the central C–C bond. The most stable conformer is the anti form, where the two methyl groups are as far apart as possible. The least stable is the fully eclipsed form, where the methyl groups are aligned.
Anti Conformer: Methyl groups are 180° apart, minimizing steric strain.
Gauche Conformer: Methyl groups are 60° apart, causing some steric strain.
Fully Eclipsed Conformer: Methyl groups are aligned, maximizing steric strain.


Cycloalkanes and Their Conformations
Ring Strain and Stability
Cycloalkanes are ring-shaped alkanes. Ring strain arises from angle strain (deviation from ideal bond angles) and torsional strain (eclipsing interactions). Cyclopropane and cyclobutane are less stable due to significant angle strain, while larger rings like cyclohexane adopt non-planar conformations to minimize strain.
Angle Strain: Caused by bond angles deviating from the ideal tetrahedral angle (109.5°).
Torsional Strain: Caused by eclipsed bonds within the ring.
Non-planar Conformations: Cyclohexane adopts a chair conformation to minimize strain.
Conformations of Cyclohexane
Cyclohexane exists primarily in the chair conformation, which is the most stable due to minimized angle and torsional strain. Other conformations include the boat, twist-boat, and half-chair, which are less stable.
Chair Conformation: All bonds are staggered, minimizing strain.
Boat Conformation: Some bonds are eclipsed, and flagpole hydrogens cause steric strain.
Twist-Boat and Half-Chair: Intermediate conformations with higher energy.



Axial and Equatorial Positions
In the chair conformation, substituents can occupy axial (vertical) or equatorial (outward) positions. Equatorial positions are preferred for bulky groups due to reduced steric interactions.
Axial Position: Points perpendicular to the ring; more prone to steric interactions.
Equatorial Position: Points outward from the ring; minimizes steric interactions.
Ring Flip: Interconversion between chair conformers swaps axial and equatorial positions.
1,3-Diaxial Interactions
Bulky substituents on cyclohexane prefer the equatorial position to avoid steric repulsion with axial hydrogens on the same side of the ring. This effect is called the 1,3-diaxial interaction.
1,3-Diaxial Interaction: Steric effect between substituents and axial hydrogens on carbons 1 and 3.
Equatorial Preference: Larger groups have a greater preference for the equatorial position.
Cis vs. Trans Isomers: Cis isomers have substituents on the same side; trans isomers have them on opposite sides.
Example: In methylcyclohexane, the methyl group prefers the equatorial position for maximum stability.
Summary Table: Classification of Alkyl Halides, Alcohols, and Amines
Type | Primary | Secondary | Tertiary | Quaternary |
|---|---|---|---|---|
Alkyl Halide | R–X (X attached to 1° C) | R2–X (X attached to 2° C) | R3–X (X attached to 3° C) | R4–X (X attached to 4° C) |
Alcohol | R–OH (OH attached to 1° C) | R2–OH (OH attached to 2° C) | R3–OH (OH attached to 3° C) | — |
Amine | R–NH2 | R2–NH | R3–N | R4–N+ |
Additional info: The notes expand on the original content by providing systematic explanations, definitions, and examples for each topic, ensuring completeness and academic quality for exam preparation.
