 Back
BackOrganic Chemistry Exam 2 Review – Nomenclature, Isomerism, and Physical Properties
Study Guide - Smart Notes

Q9. What is the relationship between the following compounds?
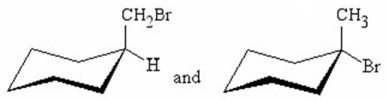
Background
Topic: Stereochemistry and Isomerism
This question tests your understanding of the different types of isomeric relationships in organic chemistry, such as constitutional isomers, conformers, enantiomers, diastereomers, and identical compounds. Recognizing these relationships is crucial for understanding molecular structure and reactivity.
Key Terms and Concepts:
Constitutional Isomers: Compounds with the same molecular formula but different connectivity of atoms.
Conformers (Conformational Isomers): Different spatial arrangements of a molecule that can be converted by rotation around single bonds.
Configurational Isomers: Isomers that cannot be interconverted by rotation around single bonds (includes enantiomers and diastereomers).
Enantiomers: Non-superimposable mirror images.
Diastereomers: Stereoisomers that are not mirror images.
Step-by-Step Guidance
Carefully examine the two cyclohexane chair structures. Identify the substituents on each ring and their positions (axial/equatorial, up/down).
Determine if the two structures have the same connectivity (i.e., are the same compound or constitutional isomers).
Check if the two structures can be interconverted by simple rotation around single bonds (conformers), or if they are different due to the arrangement of groups in space (configurational isomers).
Assess whether the two molecules are non-superimposable mirror images (enantiomers) or not. Consider the presence of chiral centers and their configurations.
Decide if the relationship is best described as identical, constitutional isomers, conformers, configurational isomers, enantiomers, or diastereomers based on your analysis.
Try solving on your own before revealing the answer!
Final Answer: Diastereomers
The two compounds are diastereomers. They are stereoisomers that are not mirror images of each other, as they differ at one stereocenter but not all. This is evident from the different substituents and their spatial arrangements on the cyclohexane rings.
