 Back
BackOrganic Chemistry I – Structure, Bonding, and Acids/Bases
Study Guide - Smart Notes

Structure and Bonding
Schematic View of an Atom
Atoms consist of a dense, positively charged nucleus surrounded by negatively charged electrons. The electron density increases as one approaches the nucleus, and most of the atom's mass is concentrated in the nucleus. Electron-density surfaces help visualize where electrons are most likely to be found.

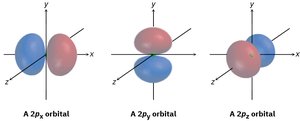
Atomic Orbitals: s, p, and d
Atomic orbitals describe regions in space where electrons are likely to be found. The s orbital is spherical, p orbitals are dumbbell-shaped, and d orbitals are cloverleaf-shaped. These shapes influence how atoms bond and interact in molecules.
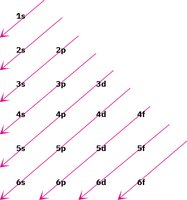
Aufbau Principle and Electron Configuration
The Aufbau principle states that electrons fill atomic orbitals in order of increasing energy, starting with the lowest energy orbital. This principle helps determine the electron configuration of atoms, which is essential for understanding chemical bonding.
Energy Levels of Electrons in an Atom
Electrons occupy energy levels (shells) around the nucleus. The first shell holds up to 2 electrons, the second up to 8, and the third up to 18. Each shell consists of subshells (s, p, d, f) with specific numbers of orbitals.
Bonding and Molecular Structure
Organic molecules are composed mainly of carbon, hydrogen, oxygen, and nitrogen. The way these atoms bond determines the structure and properties of organic compounds.
Line-Bond and Electron-Dot Structures
Organic chemists use line-bond structures (also called skeletal structures) and electron-dot structures (Lewis structures) to represent molecules. Line-bond structures show bonds as lines, while electron-dot structures show all valence electrons.
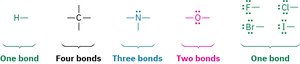
Predicting Number of Bonds
The number of bonds an atom forms is related to its group in the periodic table. For example, carbon forms four bonds, nitrogen three, oxygen two, and hydrogen one.

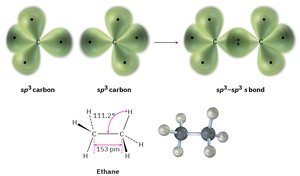
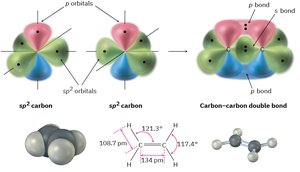
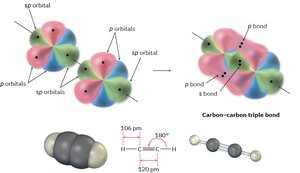
Valence Bond Theory and Sigma Bonds
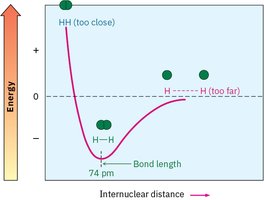
Valence bond theory explains how atomic orbitals overlap to form bonds. A sigma (σ) bond is formed by the head-on overlap of orbitals, such as in the H2 molecule. The bond has cylindrical symmetry around the bond axis.
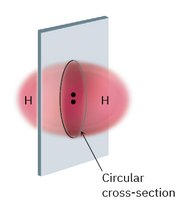
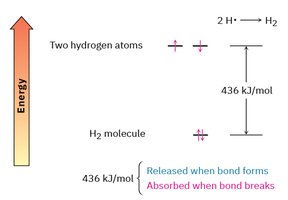
Bond Energy and Stability
Bond formation releases energy, making molecules more stable. For example, the H–H bond in H2 releases 436 kJ/mol when formed. Breaking the bond requires the same amount of energy.
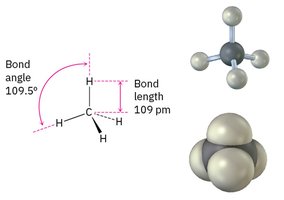
Hybridization and Molecular Geometry
Hybridization describes the mixing of atomic orbitals to form new, equivalent hybrid orbitals. The type of hybridization determines molecular geometry:
sp3 hybridization: Tetrahedral geometry, 109.5° bond angles (e.g., methane, CH4).
sp2 hybridization: Trigonal planar geometry, 120° bond angles (e.g., ethylene, C2H4).
sp hybridization: Linear geometry, 180° bond angles (e.g., acetylene, C2H2).
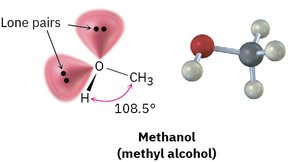
Lone Pairs
Lone pairs are nonbonding pairs of electrons localized on an atom. They influence molecular shape and reactivity.
Molecular Orbitals
Molecular orbital theory describes how atomic orbitals combine to form molecular orbitals. In H2, two 1s orbitals combine to form a bonding (lower energy) and an antibonding (higher energy) molecular orbital. Electrons fill the bonding orbital first.
Polar Covalent Bonds, Electronegativity, and Acids/Bases
Electronegativity
Electronegativity is the ability of an atom to attract electrons in a bond. Differences in electronegativity between atoms lead to bond polarity.
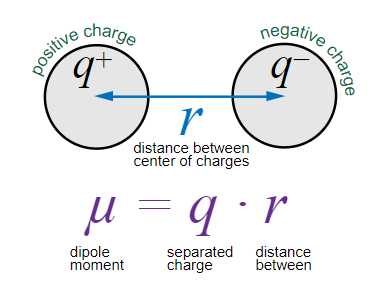
Dipole Moment and Molecular Polarity
A dipole moment (μ) is a measure of bond polarity, calculated as the product of the separated charge (q) and the distance (r) between charges: . Molecules with polar bonds may have an overall dipole moment if their shape does not cancel the bond dipoles.
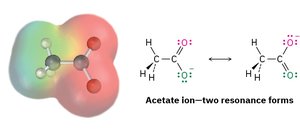
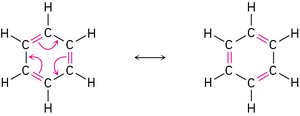
Resonance Structures
Some molecules cannot be represented by a single Lewis structure. Resonance structures are different ways of arranging electrons (not atoms) to represent the same molecule. The true structure is a resonance hybrid, which is more stable than any individual resonance form.
Acids and Bases: Brønsted-Lowry and Lewis Definitions
Brønsted-Lowry acids donate protons (H+), and bases accept protons. Lewis acids accept electron pairs, while Lewis bases donate electron pairs. Acid-base reactions involve the transfer of protons or electron pairs.
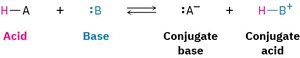
Conjugate Acid-Base Pairs
When an acid donates a proton, it forms its conjugate base. When a base accepts a proton, it forms its conjugate acid. The strength of an acid is inversely related to the strength of its conjugate base.
Acidity Constant (Ka) and pKa
The acid dissociation constant () measures the strength of an acid in water:
The pKa is the negative logarithm of : . Lower pKa values indicate stronger acids.
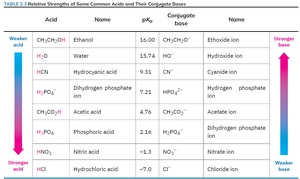
Relative Strengths of Acids and Bases
Acid and base strengths can be compared using pKa values. The direction of acid-base reactions can be predicted: reactions favor the formation of the weaker acid and base (higher pKa for acid, lower for base).
Acid | Name | pKa | Conjugate base | Name |
|---|---|---|---|---|
CH3CH2OH | Ethanol | 16.00 | CH3CH2O− | Ethoxide ion |
H2O | Water | 15.74 | HO− | Hydroxide ion |
HCN | Hydrocyanic acid | 9.31 | CN− | Cyanide ion |
H2PO4− | Dihydrogen phosphate ion | 7.21 | HPO42− | Hydrogen phosphate ion |
CH3CO2H | Acetic acid | 4.76 | CH3CO2− | Acetate ion |
H3PO4 | Phosphoric acid | 2.15 | H2PO4− | Dihydrogen phosphate ion |
HNO3 | Nitric acid | −1.3 | NO3− | Nitrate ion |
HCl | Hydrochloric acid | −7.0 | Cl− | Chloride ion |
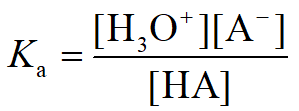
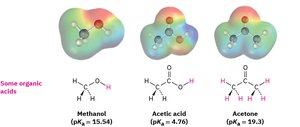
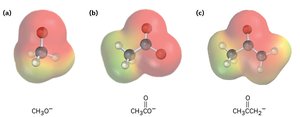
Examples of Organic Acids and Bases
Common organic acids include methanol, acetic acid, and acetone. Their conjugate bases are stabilized by electronegative atoms and resonance.
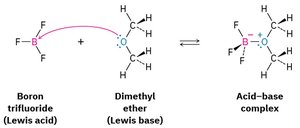
Lewis Acids and Bases
Lewis acids are electron pair acceptors, and Lewis bases are electron pair donors. Many organic reactions involve Lewis acid-base interactions, such as the reaction between boron trifluoride and dimethyl ether.

Intermolecular Forces
Intermolecular forces include dipole-dipole interactions, dispersion forces, and hydrogen bonds. These forces affect boiling points, solubility, and other physical properties of organic molecules.

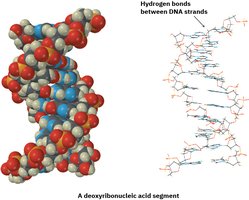
Hydrogen Bonding
Hydrogen bonds are strong dipole-dipole attractions between hydrogen attached to N, O, or F and a lone pair on another N, O, or F atom. Hydrogen bonding is crucial in biological molecules like DNA and proteins.
Hydrophilic and Hydrophobic Interactions
Hydrophilic molecules interact well with water due to polarity or hydrogen bonding, while hydrophobic molecules do not. This distinction is important in biological systems, such as cell membranes and protein folding.
