 Back
BackOrganic Chemistry Stereochemistry and Reaction Mechanisms Study Guide
Study Guide - Smart Notes

Q1. Assign R/S configuration for each chiral center. Determine which molecules are enantiomers and/or diastereomers.
Background
Topic: Stereochemistry (Chirality, Enantiomers, Diastereomers)
This question tests your ability to assign absolute configuration (R or S) to chiral centers using the Cahn-Ingold-Prelog priority rules, and to compare molecules to determine if they are enantiomers (non-superimposable mirror images) or diastereomers (stereoisomers that are not mirror images).
Key Terms and Formulas
Chiral Center: A carbon atom bonded to four different groups.
R/S Configuration: Assign priorities to the four groups, orient the lowest priority away, and determine if the sequence 1-2-3 is clockwise (R) or counterclockwise (S).
Enantiomers: Stereoisomers that are non-superimposable mirror images.
Diastereomers: Stereoisomers that are not mirror images.
Step-by-Step Guidance
For each chiral center, assign priorities (1 = highest, 4 = lowest) to the four substituents based on atomic number. If there is a tie, move to the next atom along the chain.
Orient the molecule so that the lowest priority group (4) is pointing away from you (on a dashed wedge or vertical line in a Fischer projection).
Trace a path from priority 1 → 2 → 3. If the path is clockwise, assign R; if counterclockwise, assign S.
Repeat for each chiral center in all molecules.
Compare the configurations of each molecule. Molecules with all opposite configurations are enantiomers; those with some but not all centers different are diastereomers.
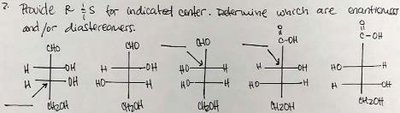
Try solving on your own before revealing the answer!
Q2. Circle the most stable carbocation.
Background
Topic: Carbocation Stability
This question tests your understanding of carbocation stability, which depends on factors such as alkyl substitution (tertiary > secondary > primary), resonance stabilization, and inductive effects.
Key Terms and Concepts
Carbocation: A positively charged carbon atom with only six electrons in its valence shell.
Stability Order: Tertiary > Secondary > Primary > Methyl; resonance and hyperconjugation increase stability.
Step-by-Step Guidance
Identify the type of carbocation (primary, secondary, tertiary) for each structure.
Check for resonance stabilization (delocalization of the positive charge).
Consider any inductive effects from nearby electronegative atoms or groups.
Compare the stability of each carbocation based on these factors.
Try solving on your own before revealing the answer!
Q3. Identify the more stable alkene (if product is requested): 1-butene vs. 2-butene.
Background
Topic: Alkene Stability
This question tests your knowledge of alkene stability, which is influenced by substitution (more substituted alkenes are more stable) and possible conjugation.
Key Terms and Concepts
Alkene: A hydrocarbon containing a carbon-carbon double bond.
Stability Order: Tetrasubstituted > Trisubstituted > Disubstituted > Monosubstituted.
Step-by-Step Guidance
Draw the structures of 1-butene and 2-butene.
Count the number of alkyl groups attached to the double-bonded carbons in each alkene.
Determine which alkene is more substituted.
Recall that more substituted alkenes are generally more stable due to hyperconjugation and alkyl group electron donation.
Try solving on your own before revealing the answer!
Q4. Complete the following reactions; show stereochemistry when needed.
Background
Topic: Organic Reaction Mechanisms and Stereochemistry
This question tests your ability to predict the products of organic reactions, including addition, elimination, and substitution reactions, and to indicate stereochemistry (cis/trans, R/S, or wedge/dash) when relevant.
Key Terms and Concepts
Reaction Mechanism: The step-by-step sequence of elementary reactions by which overall chemical change occurs.
Stereochemistry: The study of the spatial arrangement of atoms in molecules and its effect on chemical reactions.
Step-by-Step Guidance
Identify the functional groups and reagents in each reaction.
Recall the mechanism for each reaction (e.g., addition of HBr to an alkene, bromination, hydration, etc.).
Draw the expected product(s), paying attention to regiochemistry (Markovnikov/anti-Markovnikov) and stereochemistry (cis/trans, R/S, or wedge/dash as appropriate).
If a chiral center is formed, indicate the possible stereoisomers.

Try solving on your own before revealing the answer!
