 Back
BackOther Pericyclic Reactions and Benzene: Aromaticity, Nomenclature, and Reactivity
Study Guide - Smart Notes

Other Pericyclic Reactions and Benzene
Introduction to Pericyclic Reactions and Aromaticity
Pericyclic reactions are a class of organic reactions that proceed via concerted cyclic rearrangements of bonding electrons. This module expands on pericyclic reactions and introduces the concept of aromaticity, focusing on the stability and reactivity of benzene and its derivatives. Key learning objectives include predicting products of cycloaddition, electrocyclic, and sigmatropic reactions, understanding aromaticity, and applying nomenclature rules for benzene derivatives.
Pericyclic Reactions
Cycloaddition Reactions
Cycloaddition reactions involve the formation of a ring by the concerted interaction of two or more π systems. The most common example is the Diels-Alder reaction (a [4+2] cycloaddition), but other types such as [2+2] and [4+4] are also possible under specific conditions.
Suprafacial and Antarafacial Interactions: In thermal cycloadditions, symmetry-allowed reactions typically proceed via suprafacial interactions on both components. For [2+2] cycloadditions, thermal reactions are forbidden due to symmetry, but photochemical activation can allow the reaction.
Example: Drawing the MO diagram for ethene shows that a thermal [2+2] cycloaddition is forbidden due to the symmetry mismatch of the interacting orbitals.
Electrocyclic Reactions
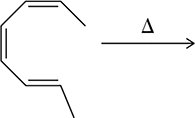
Electrocyclic reactions involve the conversion of a π system to a σ bond (or vice versa) through a concerted process. The stereochemistry of the product depends on whether the reaction is thermally or photochemically induced.
Thermal vs. Photochemical Conditions: Under thermal conditions, the reaction follows the Woodward-Hoffmann rules, leading to conrotatory or disrotatory ring closure depending on the number of π electrons.
Example: The electrocyclic closure of (2Z,4E)-hexa-2,4-diene yields different stereochemical products under heat and light.
Sigmatropic Rearrangements
Sigmatropic rearrangements are pericyclic reactions where a σ bond adjacent to one or more π systems migrates across the molecule. The most common are [1,5]- and [3,3]-sigmatropic shifts (e.g., the Cope and Claisen rearrangements).
Chair-like Transition State: The stereochemical outcome can often be rationalized by considering a chair-like transition state.
Classification: Sigmatropic rearrangements are classified by the number of atoms involved in the migration (e.g., [3,3] or [1,5]).
Aromaticity and Benzene
Discovery and Structure of Benzene
Benzene is a planar, cyclic molecule with six π electrons delocalized over six carbon atoms. Its unusual stability is attributed to aromaticity, a concept defined by specific electronic and structural criteria.
Hückel's Rule: A molecule is aromatic if it is cyclic, planar, fully conjugated, and contains 4n+2 π electrons (where n is an integer).
Example: Benzene (n=1) has 6 π electrons and is aromatic.
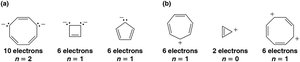
Criteria for Aromatic, Antiaromatic, and Nonaromatic Compounds
Compounds are classified based on their electronic structure:
Aromatic: Cyclic, planar, fully conjugated, and 4n+2 π electrons.
Antiaromatic: Cyclic, planar, fully conjugated, and 4n π electrons (n is an integer), leading to destabilization.
Nonaromatic: Compounds that do not meet the criteria for aromaticity or antiaromaticity (e.g., not planar or not fully conjugated).
Molecular Orbitals of Aromatic and Antiaromatic Compounds
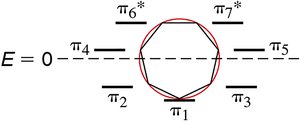
The stability of aromatic compounds can be explained using molecular orbital (MO) theory. Aromatic compounds have completely filled bonding π orbitals, while antiaromatic compounds have unpaired electrons in degenerate orbitals, leading to instability.
Frost Circles: Frost diagrams are used to visualize the relative energies of π molecular orbitals in cyclic systems.
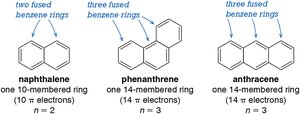
Polycyclic Aromatic Hydrocarbons
Polycyclic aromatic hydrocarbons (PAHs) are compounds with multiple fused benzene rings. Their aromaticity and stability depend on the number and arrangement of rings.
Examples: Naphthalene, anthracene, and phenanthrene are common PAHs, each with unique electronic properties.
Nomenclature of Benzene Derivatives
IUPAC Nomenclature
Benzene derivatives are named by identifying the substituents and their positions on the ring. The parent name is 'benzene,' and substituents are given the lowest possible locants.
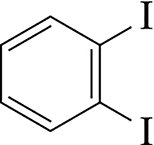
Example: 1,2-diiodobenzene, 3-chlorobenzoic acid, 4-isopropylphenol.
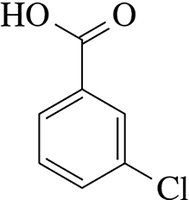
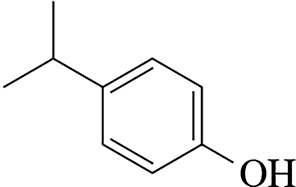
Ortho, Meta, and Para Nomenclature
The ortho (o-), meta (m-), and para (p-) system is used for disubstituted benzenes to indicate relative positions:
Ortho (o-): Substituents at positions 1,2
Meta (m-): Substituents at positions 1,3
Para (p-): Substituents at positions 1,4
Effects of Aromaticity on Reactivity
Aromaticity and Acidity/Basicity
Aromaticity can significantly affect the acidity and basicity of compounds. For example, the presence of an aromatic ring can stabilize a conjugate base via delocalization of negative charge, increasing acidity.
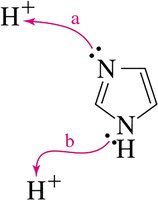
Example: Imidazole is a heteroaromatic base; the basicity of its nitrogens depends on their involvement in the aromatic π system.
Aromaticity and SN1 Reaction Rates
The stability of carbocation intermediates in SN1 reactions is enhanced if the carbocation is aromatic, leading to faster reaction rates.
Practice Problems and Applications
Classifying Compounds
Given a structure, classify it as aromatic, antiaromatic, or nonaromatic based on the criteria discussed above.
Frost Diagrams for Aromatic and Antiaromatic Systems
Draw Frost diagrams for various cyclic systems to determine their aromatic or antiaromatic character by filling in the appropriate number of π electrons.
Comparing Basicity and Nucleophilicity
Compare the basicity of nitrogens in heterocycles (e.g., imidazole, pyrrole, pyrrolidine) based on their involvement in aromaticity and electron delocalization.
Summary Table: Aromaticity Classification
Compound | π Electrons | Planarity | Classification |
|---|---|---|---|
Benzene | 6 | Yes | Aromatic |
Cyclobutadiene | 4 | Yes | Antiaromatic |
Cyclooctatetraene | 8 | No | Nonaromatic |
Cyclopentadienyl anion | 6 | Yes | Aromatic |
Cyclopropenyl cation | 2 | Yes | Aromatic |
Conclusion
This module provides a comprehensive overview of pericyclic reactions and aromaticity, including the prediction of reaction products, the application of nomenclature rules, and the analysis of aromatic stabilization. Mastery of these concepts is essential for understanding the reactivity and properties of organic molecules.
